One kilogram of water vapor at 200 kPa fills the 1.1989-m 3 left chamber of a partitioned
Question:
One kilogram of water vapor at 200 kPa fills the 1.1989-m3 left chamber of a partitioned system shown in Fig. P3–37.
The right chamber has twice the volume of the left and is initially evacuated. Determine the pressure of the water after the partition has been removed and enough heat has been transferred so that the temperature of the water is 3°C.
Transcribed Image Text:
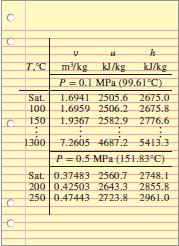
T.C m/kg K/kg P 0.1 MPa (99.61°C) KJ/kg Sat. T.6941 2505.6 2675.0 1.6959 2506.2 2675.8 100 150 1.9367 2582.9 2776.6 1300 7.2605 4687.2 5413.3 P=0.5 MPa (151.83°C) Sat. 0.37483 2560.7 2748.1 200 0.42503 2643.3 2855.8 250 0.47443 2723.8 2961.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The initial specific volume is At the final state the water occ...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Tire City Inc. Mr. Abdullah, Chief Financial Officer (CFO) of Tire City Inc ('TCI') has a meeting with their bank later in the week. At the meeting he needs to present a request to the bank for...
-
During the current year, Rowan sells her entire interest in Concord Corporation common stock for $15,000. She is the sole shareholder, and originally organized the corporation several years ago by...
-
The radiator of a steam heating system has a volume of 20 L and is filled with superheated water vapor at 200 kPa and 200C. At this moment both the inlet and the exit valves to the radiator are...
-
If the current spot rate is 2.10 (HC/FC) and the government undertakes policies to depreciate the currency 16%, what is the new spot rate? Note: 2.10 * 1.16 is not the right formula. Based on this,...
-
A sporting goods manufacturing company wanted to compare the distance traveled by golf balls produced using each of four different designs. Ten balls of each design were manufactured and brought to...
-
Ethical Choice / Decision: Situation Fyre Festival was a heavily marketed event that didn't follow through what their advertisements promised. One of the key decision makers was Billy McFarland. He...
-
As shown in Fig. 5-7p, a positive lens with focal length \(f\) is placed in contact with a pinhole screen in a Young's experiment. The lens and pinhole plane are at distance \(z_{1}\) from the source...
-
Walden Green Company purchased land containing an estimated 4,000,000 tons of ore for $16,000,000. The land will be worth $2,400,000 without the ore after 8 years of active mining. Although the...
-
Leadership is an integral element in any job, regardless of the work title. However, it is important to recognize that leadership is not just one single skill; instead, success in leadership depends...
-
Eric Krassows firm is about to bid on a new radar system. Although the product uses new technology, Krassow believes that a learning rate of 75% is appropriate. The first unit is expected to take 700...
-
One kilogram of R-134a fills a 0.14-m 3 weighted pistoncylinder device at a temperature of 26.4C. The container is now heated until the temperature is 100C. Determine the final volume of the R-134a.
-
A rigid tank with a volume of 1.8 m 3 contains 40 kg of saturated liquid vapor mixture of water at 90C. Now the water is slowly heated. Determine the temperature at which the liquid in the tank is...
-
In the following cases, check that x = c is a critical point and use Exercise 75 to determine whether (c) is a local minimum or a local maximum. Data From Exercise 75 The Second Derivative Test for...
-
A 300 kg satellite is in circular orbit around the earth ( M = 5.98 1024 kg) at a speed of 6200 m/s. a. Find the radius of the satellite's orbit. Show your work. (3 marks) b. Find the gravitational...
-
A 1,700 word memo to demonstrate your skill at applying relevant law to the scenario. You are an articling student who is working for defence counsel Dewey, Cheatem and Howe who have been hired by...
-
Walberg Associates, antique dealers, purchased goods for $37,500. Terms of the purchase were FOB shipping point, and the cost of transporting the goods to Walberg Associates's warehouse was $1,200....
-
For a special whole life insurance issued to (40), a benefit of $1,000 is payable at the moment of death if the insured dies within the first 10 policy years; a benefit of $2,000 is payable at the...
-
5. A billiard ball of mass 0.15 kg is rolling north at 3.5 m/s when it collides with a stationary golf ball of mass 0.050 kg. The collision is a glancing one and the billiard ball has a velocity of...
-
Blue Mountain Products manufactures and sells a variety of camping products. Recently, the company opened a new plant to manufacture a lightweight, self-standing tent. Cost and sales data for the...
-
The first national bank pays a 4% interest rate compound continuously. The effective annual rate paid by the bank is __________. a. 4.16% b. 4.20% c. 4.08% d. 4.12%
-
A 1-m3 tank contains 2.841 kg of steam at 0.6 MPa. Determine the temperature of the steam, using (a) The idealgas equation, (b) The van der Waals equation, and (c) The steam tables.
-
Reconsider Prob. 3-100. Solve the problem using EES (or other) software. Again using the EES, compare the temperature of water for the three cases at constant specific volume over the pressure range...
-
During a hot summer day at the beach when the air temperature is 30oC, someone claims the vapor pressure in the air to be 5.2 kPa. Is this claim reasonable?
-
www The sugar cane industry is a big industry on Hawaii. Raw sugar cane is first cut from the fields, then chopped and shredded. The raw cane contains 15 wt% sucrose, 25 wt% solids, and water, along...
-
What does a manager who subscribes to Theory X believes ? Explain thoroughly.
-
Keri, in forming a new corporation, transfers land to the corporation in exchange for 100 percent of the stock of the corporation. Keri's basis in the land is $295,000, and the corporation assumes a...

Study smarter with the SolutionInn App


