Reconsider Prob. 1443. Determine the adiabatic saturation temperature of the humid air. Data From Prob. 1443. Atmospheric
Question:
Reconsider Prob. 14–43. Determine the adiabatic saturation temperature of the humid air.
Data From Prob. 14–43.
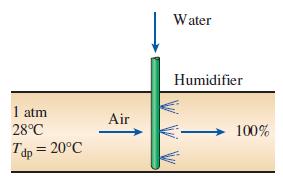
Atmospheric air at a pressure of 1 atm and dry-bulb temperature of 28°C has a dew-point temperature of 20°C. Using the psychrometric chart.
Transcribed Image Text:
Water Humidifier 1 atm 28°C Air 100% Tdp = 20°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
The pressure temperature and wetbulb temperature of air are specified The adiabatic sa...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Determine the adiabatic saturation temperature of the humid air in Prob. 14-41. Prob. 14-41. Atmospheric air at a pressure of 1 atm and drybulb temperature of 28oC has a wet-bulb temperature of 20oC....
-
Determine the adiabatic saturation temperature of the humid air in Prob. 14-43E. Prob. 14-43E Atmospheric air at a pressure of 1 atm and drybulb temperature of 90oF has a dew-point temperature of...
-
Reconsider Prob. 1439E. Determine the adiabatic saturation temperature of the humid air. Data From Prob. 1439E: Atmospheric air at a pressure of 1 atm and dry-bulb temperature of 90F has a wet-bulb...
-
Presented below is information related to LeBron James Manufacturing Corporation. Instructions (a) Compute the rate of depreciation per year to be applied to the plant assets under the composite...
-
The number 22/7 is similar to TT in the sense that they both require an infinite number of decimal places. However, 22/7 is a rational number because it can be expressed as the ratio of two integers,...
-
A consolidated income statement for 2018 and comparative consolidated balance sheets for 2017 and 2018 for P Company and its 80% owned subsidiary follow: Other information: 1. Equipment depreciation...
-
Water flows from a two-dimensional open channel and is diverted by an inclined plate as illustrated in Fig. P5.78. When the velocity at section (1) is \(10 \mathrm{ft} / \mathrm{s}\), what horizontal...
-
George Company purchased land for use as its corporate headquarters. A small factory that was on the land when it was purchased was torn down before construction of the office building began....
-
Write an assembly code to compute the following expression: y=(AC)+(B+2) where y is 32-bit regist er, and A, C and B are 16-bit memory variables and A-14, C = 10 and B = -1 Note: You are only allowed...
-
Audit standards distinguish auditors responsibility for planning procedures for detecting noncompliance with laws and regulations having a direct effect on financial statements versus planning...
-
What does a modern air-conditioning system do besides heating or cooling the air?
-
Atmospheric air at a pressure of 1 atm and dry-bulb temperature of 90F has a wet-bulb temperature of 85F. Using the psychrometric chart, determine (a) The relative humidity (b) The humidity ratio (c)...
-
For the cash flows shown, determine the present worth in year 0, if the interest rate is 12% peryear. Year Cash Flow, S13 13 13 13 16 19 22 25 28 31 12 3 4 5 6 7 8 9 10
-
For the system given below, an observer is to be designed to estimate the state variables. Select the observer gain and write the equations describing the observer dynamics. Also develop the block...
-
Use Nyquist criterion to determine range of \(\mathrm{K}\) for closed loop stability of the open loop function with unity feedback given by: \[ \mathrm{G}(s)=\frac{\mathrm{K} e^{-0.8 s}}{(s+1)} \]
-
Sketch the root locus for K K ranging from 0 to for a system with product G ( s ) H ( s ) = K ( s 4 ) 2 ( s + 5 ) ( s 2 + 4 s + 10 ) G ( s ) H ( s ) = K ( s 4 ) 2 ( s + 5 ) ( s 2 + 4 s + 10 )...
-
The first element of each of the rows of a Routh-Hurwitz stability test showed the sign as follows The number of roots of the system lying in the right half of \(s\)-plane is (a) 2 (b) 3 (c) 4 (d) 5...
-
Consider the system shown in Fig. P 5.9(a). (a) Sketch the root locus as \(\mathrm{z}\) varies from 0 to \(\infty\). (b) Determine the value of \(\mathrm{z}\) so that damping ratio of dominant...
-
(a) By taking the slope of the curve in Figure 2.60, verify that the velocity of the jet car is 115 m/s at t = 20 s. (b) By taking the slope of the curve at any point in Figure 2.61, verify that the...
-
State whether each statement is true or false. If false, give a reason. {purple, green, yellow} = {green, pink, yellow}
-
The volumetric analysis of a mixture of gases is 30 percent oxygen, 40 percent nitrogen, 10 percent carbon dioxide, and 20 percent methane. This mixture flows through a 1.6-cm-diameter pipe at 8000...
-
A rigid tank contains 1 lbmol of argon gas at 400 R and 750 psia. A valve is now opened, and 3 lbmol of N2 gas is allowed to enter the tank at 340 R and 1200 psia. The final mixture temperature is...
-
The volumetric analysis of mixture of gases is 30 percent oxygen, 40 percent nitrogen, 10 percent carbon dioxide, and 20 percent methane. This mixture is heated from 20oC to 200oC while flowing...
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
11 The APRN unit director is working at an agency that has a high incidence of medication errors, specifically with heparin. One recent error had a patient receive twice the prescribed dose. Which...

Study smarter with the SolutionInn App


