Repeat Prob. 128 for helium. Data From Q#8: Consider air at 350 K and 0.75 m 3
Question:
Repeat Prob. 12–8 for helium.
Data From Q#8:
Consider air at 350 K and 0.75 m3/kg. Using Eq. 12–3, determine the change in pressure corresponding to an increase of
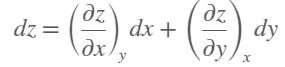
(a) 1 percent in temperature at constant specific volume
(b) 1 percent in specific volume at constant temperature
(c) 1 percent in both the temperature and specific volume.
Transcribed Image Text:
dz dz dx + dy = %3D дх. y ду,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Helium at a specified temperature and specific volume is considered ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Repeat Problem 12-5 for helium. Problem 12-5 Consider air at 350 K and 0.75 m3/kg. Using Eq. 12-3, determine the change in pressure corresponding to an increase of (a) 1 percent in temperature at...
-
Consider air at 350 K and 0.75 m3/kg. Using Eq. 12-3, Determine the change in pressure corresponding to an increase of (a) 1 percent in temperature at constant specific volume. (b) 1 percent in...
-
Consider air at 400 K and 0.90 m3/kg. Using Eq. 123, determine the change in pressure corresponding to an increase of (a) 1 percent in temperature at constant specific volume, (b) 1 percent in...
-
Jacomo Companys output for the current period was assigned a $300,000 standard direct materials cost. The direct materials variances included a $44,000 favorable price variance and a $6,000 favorable...
-
Find the regression equation; unless the problem suggests otherwise, let the first variable be the independent (x) variable. Caution: When finding predicted values, be sure to follow the prediction...
-
13. Suppose you are a currency trader in South Korea. The exchange rate for the Korean won relative to the U.S. dollar is 1,114.95. This figure is Korean won (KRW) per U.S. dollar (USD). Assume the...
-
The velocity potential for a spiral vortex flow is given by \(\phi=\) \((\Gamma / 2 \pi) \theta-(m / 2 \pi) \ln r\), where \(\Gamma\) and \(m\) are constants. Show that the angle, \(\alpha\), between...
-
Clean-All, Inc., sells washing machines with a three-year warranty. In the past Clean-All has found that in the year after sale, warranty costs have been 3% of sales; in the second year after sale,...
-
A 1.3 kg book is lying on a 0.75 m -high table. You pick it up and place it on a bookshelf 2.3 m above the floor. During this process, how much work does gravity do on the book? During this process,...
-
An ISP is granted the block 16.12.64.0/20. The ISP needs to allocate addresses for 8 organizations, each with 256 addresses. a. Find the number and range of addresses in the ISP block. b. Find the...
-
Consider air at 350 K and 0.75 m 3 /kg. Using Eq. 123, determine the change in pressure corresponding to an increase of (a) 1 percent in temperature at constant specific volume (b) 1 percent in...
-
Show how you would evaluate T, v, u, a, and g from the thermodynamic function h = h(s, P).
-
Why is knowledge of cost behavior important for managerial decision making? Give an example to illustrate your answer.
-
Grant and Herd are in partnership sharing profits and losses in the ratio 3 to 2. The following information relates to the year to 31 December 2011: 1 The partnership agreement allows for Herd to be...
-
It has been suggested that CSR from an Islamic perspective has a wider focus. Discuss.
-
How are social networks structured?
-
Choose a social game to play. As you interact with the game, keep a journal of your experience. In particular, note the advertising and branded components and your reactions to them. How does your...
-
How can entertainment brands leverage social TV?
-
Financial information for Cao Inc. follows. Instructions (a) Calculate the following ratios or relationships of Cao Inc. Assume that the ending account balances are representative unless the...
-
Digital Fruit is financed solely by common stock and has outstanding 25 million shares with a market price of $10 a share. It now announces that it intends to issue $160 million of debt and to use...
-
Using EES (or other) software, determine how much the thermal efficiency of the cycle in Prob. 10-22 would change if there were a 50 kPa pressure drop across the boiler.
-
The net work output and the thermal efficiency for the Carnot and the simple ideal Rankine cycles with steam as the working fluid are to be calculated and compared. Steam enters the turbine in both...
-
A binary geothermal power plant uses geothermal water at 160°C as the heat source. The cycle operates on the simple Rankine cycle with isobutane as the working fluid. Heat is transferred to the...
-
How do the resource descriptions (typing) used in the Incident Command System lead to a more effective response?
-
A company incurs $4172000 of overhead each year across three departments: Ordering and Receiving, Mixing, and Testing. The company prepares 2000 purchase orders, works 50000 mixing hours, and...
-
Hemming Company reported the following current-year purchases and sales for its only product. Date January 1 January 10 Activities Beginning inventory March 14 March 15 July 30 Sales Purchase Sales...

Study smarter with the SolutionInn App


