The equation of state of a gas is given by where a and b are constants. Use
Question:
The equation of state of a gas is given by
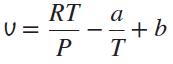
where a and b are constants. Use this equation of state to derive an equation for the Joule-Thomson coefficient inversion line.
Transcribed Image Text:
RT U = a + b T %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
The equation of state of a gas is given by An equation for the JouleThomson coefficient inv...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
The equation of state of a gas is given as (P + 10/ 2) = RuT, where the units of and P are m3/kmol and kPa, respectively. Now 0.5 kmol of this gas is expanded in a quasi-equilibrium manner from 2 to...
-
The equation of state of a gas is given as v-(P + 10/ v-2) = 5 RuT, where the units of v- and P are m3/kmol and kPa, respectively. Now 0.2 kmol of this gas is expanded in a quasi equilibrium manner...
-
What must be the beta of a portfolio with E( rP ) = 18%, if rf = 6% and E (rM) = 14%?
-
Find the regression equation; unless the problem suggests otherwise, let the first variable be the independent (x) variable. Caution: When finding predicted values, be sure to follow the prediction...
-
Write a server for a client. The client sends the weight and height for a person to the server (see Figure 31.18a). The server computes BMI (Body Mass Index) and sends back to the client a string...
-
A certain flow field is described by the stream function \[ \psi=A \theta+B r \sin \theta \] where \(A\) and \(B\) are positive constants. Determine the corresponding velocity potential and locate...
-
Sally Saia operates Double S Riding Academy, Inc. The academys primary sources of revenue are riding fees and lesson fees, which are provided on a cash basis. Sally also boards horses for owners, who...
-
The Samsung Global Code of Conduct and Business Conduct Guidelines, which direct every one of their representatives, drive their efforts to deliberately oversee consistency and moral dangers. It also...
-
Hewitt Corporation has these accounts at December 31: Common Stock, $10 par, 5,000 shares issued, $50,000; Paid-in Capital in Excess of Par Value $22,000; Retained Earnings $42,000; and Treasury...
-
What is the most general equation of state for which the Joule-Thomson coefficient is always zero?
-
What is the enthalpy departure?
-
Zak Corp. purchased depreciable assets costing $600,000 on January 2, 2020. For tax purposes, the company uses CCA in a class that has a 40% rate. Assume these assets are considered eligible...
-
What is a variable-length array definition?
-
Review the list of friends you have on Facebook. How many of your friends are weak ties and how many are strong ties? Identify the relationship bonds you share with those in your strong tie group....
-
What is social entertainment? What are the types of social entertainment?
-
How do ratings and reviews provide value for consumers and marketers?
-
What is a social media marketing tactical plan and how does it support the execution of a social media marketing strategy?
-
The incomplete income statement of Justin Corp. follows. The employee profit-sharing plan requires that 20% of all profits remaining after the deduction of the bonus and income tax be distributed to...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
Why is the combined gas-steam cycle more efficient than either of the cycles operated alone?
-
The gas-turbine portion of a combined gas-steam power plant has a pressure ratio of 16. Air enters the compressor at 300 K at a rate of 14 kg/s and is heated to 1500 K in the combustion chamber. The...
-
Consider a combined gas-steam power plant that has a net power output of 450 MW. The pressure ratio of the gas-turbine cycle is 14. Air enters the compressor at 300 K and the turbine at 1400 K. The...
-
Give an example of one piece of financial information that may be used differently for two stakeholders (users) and explain how it would be used and presented differently?
-
Complete common -size statements (balance sheets) using the data below on page 246. You will be required to create a spreadsheet in MS Excel to display the statements (balance sheets).? In a 1 to 2...
-
A sail boat in the Great South Bay travels 2 miles east and then 4 miles south to get to Fire Island. What is its distance and displacement?

Study smarter with the SolutionInn App


