Use the Gibbs function to determine the equilibrium constant of the H 2 O H 2
Question:
Use the Gibbs function to determine the equilibrium constant of the H2O ⇌ H2 + 1/2 O2 reaction at
(a) 1440 R
(b) 3960 R.
How do these compare to the equilibrium constants of Table A–28?
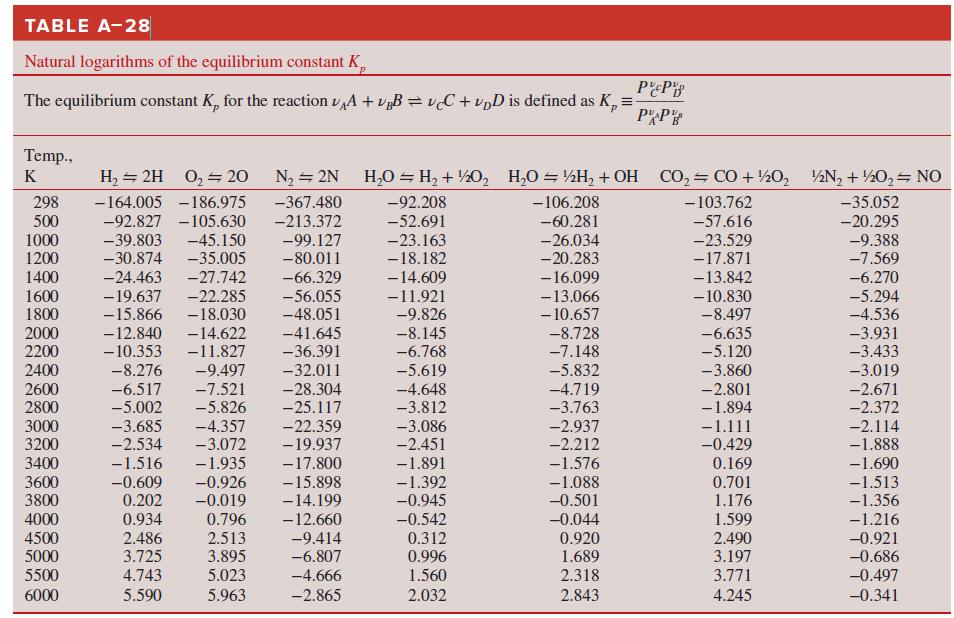
Transcribed Image Text:
TABLE A-28 Natural logarithms of the equilibrium constant K, The equilibrium constant K, for the reaction vA + v„B = vC + vpD is defined as K, = PP Temp., K H, = 2H 0, 20 N2 = 2N H,0 = H, + ½0, H,O = ½H, + OH CO, CO + ½0, ½N, + ½0, NO 298 500 1000 -164.005 -186.975 -367.480 -92.208 -106.208 - 103.762 -35.052 -92.827 -105.630 -45.150 -35.005 -27.742 -213.372 -52.691 -60.281 -57.616 -20.295 -39.803 -30.874 -99.127 -80.011 -9.388 -7.569 -23.163 -26.034 -20.283 -23.529 -17.871 -13.842 1200 -18.182 -6.270 -5.294 1400 -24.463 -66.329 -14.609 1600 1800 -19.637 -15.866 -22.285 -18.030 -14.622 - 16.099 -13.066 - 10.657 -56.055 -48.051 -11.921 -9.826 - 10.830 -8.497 -6.635 -5.120 -4.536 2000 2200 -12.840 - 10.353 -41.645 -8.145 -6.768 -8.728 -7.148 -3.931 -3.433 -11.827 -36.391 2400 -8.276 -9.497 -32.011 -5.619 -4.648 -3.812 -5.832 -3.860 -3.019 -28.304 2600 2800 -6.517 -5.002 -7.521 -5.826 -4.719 -3.763 -2.801 -1.894 -2.671 -2.372 -25.117 -22.359 3000 3200 -3.685 -2.534 -4.357 -3.072 -3.086 -2.937 -2.212 -1.111 -0.429 -2.114 -1.888 -19.937 -2.451 3400 -1.516 -1.935 -17.800 -1.891 -1.576 0.169 3600 3800 -0.609 0.202 -0.926 -0.019 -15.898 -14.199 -12.660 -1.690 -1.513 -1.356 -1.392 -0.945 -1.088 -0.501 0.701 1.176 0.796 2.513 3.895 0.934 -0.542 -0.044 0.920 1.689 4000 1.599 -1.216 4500 5000 2.486 3.725 -9.414 -6.807 0.312 2.490 3.197 -0.921 -0.686 0.996 5500 4.743 5.023 -4.666 1.560 2.318 3.771 -0.497 6000 5.590 5.963 -2.865 2.032 2.843 4.245 -0.341
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
The equilibrium constant of the reaction is to be dete...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
The equilibrium constant for the reaction H2 + at 1 atm and 1500C is given to be K. Of the reactions given below, all at 1500C, the reaction that has a different equilibrium constant is (a) H2 + 12O2...
-
The equilibrium constant for the H2 + ½ O2 H2O reaction at 1 atm and 1200 K is KP. Use this information to determine the equilibrium constant for the following reactions: (a) at l atm H, +...
-
The table below shows data (from a 2004 Bureau of the Census report) on the number of times 20- to 24-year-old men have been married. a. Verify that the mean number of times men have been married is...
-
Consider a portfolio of the following derivatives where the counterparty is an OECD bank. Derivative 8-year interest rate swap 6-month option on an equity 1-year swap on precious metals 9-month...
-
Digits (0, 1, 2,. . . ,9) are randomly selected for telephone numbers in surveys. The random variable x is the selected digit. a. Find the mean and standard deviation of x. b. Find the z score for...
-
Stellar Stores is a new company that started operations on March 1, 2021. The company has decided to use a perpetual inventory system. The following purchase transactions occurred in March: Mar. 1...
-
Describe briefly how a schlieren optical visualization system (Fig. 11.4) works. How else might density changes in a fluid flow be made visible to the eye? Fig. 11.4 Shock wave Flow Airfoil
-
Megan has her home and personal property insured under an unendorsed Homeowners 3 (special form) policy. Indicate whether each of the following losses is covered. If the loss is not covered, explain...
-
What is the role of cross-functional teams in TPM, and how do they contribute to the success of TPM initiatives? Discuss the importance of collaboration between production, maintenance, and...
-
1. Imagine that an agency or business has asked for help in gathering data about the number of sports anglers who fish off the coast of Georgia. What advice would you give about sampling? What method...
-
A mixture of ideal gases consists of the following gases by mole fraction: 10 percent CO 2 , 60 percent H 2 O, and 30 percent CO. Determine the Gibbs function of the CO in this mixture when the...
-
An inventor claims she can produce hydrogen gas by the reversible reaction 2H 2 O H 2 + O 2 . Determine the mole fractions of the hydrogen and oxygen produced when this reaction occurs at 4000 K and...
-
Given that P (A | B) = .44 and P (A and B) = .33, find P (B)
-
In some states plaintiffs who file a wrongful-death action can recover for a. loss of companionship and sexual relationship. b. monetary contributions the decedent would have made during their...
-
What is the difference between a governmental function and a proprietary function?
-
In assessing future lost wages, attorneys consult with____________ ____________ ____________ counselors who help predict what the plaintiff will be able to do and how much schooling will be required.
-
____________ ____________ value refers to the amount property can be sold for on the open market.
-
True Or False Last clear chance doctrine is available if both plaintiff and defendant are inattentive.
-
At December 31, 2017, Tres Hombres Corporation had the following shares outstanding: 10% cumulative preferred shares, 107,500 shares outstanding ........... $10,750,000 Common shares, 4,000,000...
-
As long as we can't lose any money, we have a risk-free investment." Discuss this comment. Q2: Both investing and gambling can be defined as "undertaking risk in order to earn a profit." Explain how...
-
Octane gas (C8H18) at 25oC is burned steadily with 30 percent excess air at 25oC, 1 atm, and 60 percent relative humidity. Assuming combustion is complete and adiabatic, calculate the exit...
-
Reconsider Prob. 15-75. Using EES (or other) software, investigate the effect of the relative humidity on the exit temperature of the product gases. Plot the exit temperature of the product gases as...
-
A coal from Pennsylvania has an ultimate analysis (by mass) as 84.36 percent C, 1.89 percent H2, 4.40 percent O2, 0.63 percent N2, 0.89 percent S, and 7.83 percent ash (non-combustibles) is burned in...
-
write the differences between Microprocessors and Microcomputers. compare between them and draw the block diagram for each one. B) define the interface and mention it's types with brief description....
-
Write a program that prompts the user for the constitutive parameters (relative permittivity, relative permeability, and conductivity) in medium 1 and medium 2 separated by a planar surface. You are...
-
3. Given the following circuit... A B C a. Analyze the behavior of the above circuit with a truth table. Create a column for every gate excluding NOT gates. b. What is the equivalent Boolean...

Study smarter with the SolutionInn App


