Using Daltons law, show that for a real-gas mixture of k gases, where Z is the compressibility
Question:
Using Dalton’s law, show that
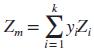
for a real-gas mixture of k gases, where Z is the compressibility factor.
Transcribed Image Text:
Z„ = EyZi %3D i=1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Using Daltons law it is to be shown that for a realgas mixture Analysis U...View the full answer

Answered By

BETHUEL RUTTO
Hi! I am a Journalism and Mass Communication graduate; I have written many academic essays, including argumentative essays, research papers, and literary analysis. I have also proofread and written reviews, summaries and analyses on already finished works. I am eager to continue writing!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
A gas turbine is operated with gases (Cp = 0.992 kJ/kg K and = 1.29) entering it at 10 bar and 1025C, and leaving it at 1 bar and 550C. Assuming adiabatic flow through the turbine, calculate the...
-
A gas mixture at 300 K and 200 kPa consists of 1 kg of CO2 and 3 kg of CH4. Determine the partial pressure of each gas and the apparent molar mass of the gas mixture.
-
A mixture of Ne and Ar gases at 350 K contains twice as many moles of Ne as of Ar and has a total mass of 50.0 g. If the density of the mixture is 4.00 g/L, what is the partial pressure of Ne?
-
Lucy, a 57-year-old widow, lost her husband in 2021. He was an active participant in the Federal Employees' Retirement System (FERS), and his FERS contributions totaled $27,000 at the time of his...
-
In the previous question, 1350 is an outlier. If we remove this value, show why the sales without outliers are normally distributed at a 0.05 level of significance.
-
Waterbahn Waterslide Company issued 25,000 ten-year, 6 percent, $100 bonds on January 1, 2017, at face value. Interest is payable each December 31. Show the accounting equation effects and prepare...
-
A pump moves water horizontally at a rate of \(0.02 \mathrm{~m}^{3} / \mathrm{s}\). Upstream of the pump where the pipe diameter is \(90 \mathrm{~mm}\), the pressure is \(120 \mathrm{kPa}\)....
-
During December, Rainey Equipment made a $600,000 credit sale. The state sales tax rate is 6% and the local sales tax rate is 1.5%. Prepare the appropriate journal entry.
-
Consider a project that requires an investment of $1700 in the first year, but will generate a NPV of $80. Calculate the profitability index value of this project.
-
Which of the following is/are the correct syntax of specifying choices for a random function: 1) random.choice(2,3,4) 2) random.choice([2,3,4]) 3) random.choice( (2,3,4)) a) None b) 1, 2, 3 are...
-
A mixture of gases consists of 1 kmol of carbon dioxide, 1 kmol of nitrogen, and 0.3 kmol of oxygen. Determine the total amount of work required to compress this mixture isothermally from 10 kPa and...
-
A 60-L rigid tank contains an ideal-gas mixture of 5 g of N 2 and 5 g of CO 2 at a specified pressure and temperature. If N 2 were separated from the mixture and stored at mixture temperature and...
-
Determine the luminous efficacy of the following lamps having comparable illumination outputs: a. A 300 W incandescent lamp that emits 5820 lm b. Two 32 W linear fluorescent lamps that emit 2950 lm...
-
Media Skills: Social Networking Joining an ongoing social media conversation, such as a comment thread on Facebook, requires a delicate touch if you work for a company that has a commercial interest...
-
Media Skills: Microblogging Busy knitters can go through a lot of yarn in a hurry, so most keep a sharp eye out for yarn sales. Youre on the marketing staff of Knitting-Warehouse, and you like to...
-
Revise these sentences to be positive rather than negative: 1. You have not filled in every section of the questionnaire we sent you. 2. Its clear that you have not understood the question. 3. It is...
-
Writing: Using Plain Language; Communication Ethics: Making Ethical Choices, Chapter 1 Your company has been a major employer in the local community for years, but shifts in the global marketplace...
-
Each of the following sentences contains at least one error in spelling, grammar, capitalization, punctuation, abbreviation, number style, word division, or vocabulary. Rewrite each sentence,...
-
Bird bones have air pockets in them to reduce their weight-this also gives them an average density significantly less than that of the bones of other animals. Suppose an ornithologist weighs a bird...
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
A reversible absorption refrigerator consists of a reversible heat engine and a reversible refrigerator. The system removes heat from a cooled space at - 15oC at a rate of 70 kW. The refrigerator...
-
An ammonia-water absorption refrigeration cycle is used to keep a space at 25oF when the ambient temperature is 70oF. Pure ammonia enters the condenser at 300 psia and 140oF at a rate of 0.04lbm/s....
-
Describe the Seebeck and the Peltier effects.
-
Example: Make vs Buy: A decision concerning whether an item should be produced internally or purchased from an outside supplier. Java J's is a coffee shop offering a variety of coffees, teas and...
-
3. An elastic material fills a cubic-shaped cavity in a rigid metal form. The properties of the material are: E = 15000 MPa v = 0.25 = 120 10-6/C (coefficient of thermal expansion) The material is...
-
Every decision has an Opportunity Cost due to the nature of scarcity, there is always a better alternative not chosen, therefore, there is always an opportunity cost. "The opportunity cost of an...

Study smarter with the SolutionInn App


