An insulated rigid tank with a volume of 0.2 m 3 contains steam at 100 kPa and
Question:
An insulated rigid tank with a volume of 0.2 m3 contains steam at 100 kPa and 400 K and a 4-cm3 ice cube at 0° C. Determine the equilibrium temperature of the water and the change in entropy during the process. For the ice,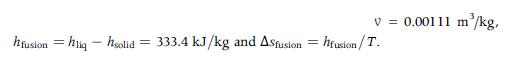
Transcribed Image Text:
v = 0.00111 m³/kg, hfusion = hliq - hsolid = 333.4 kJ/kg and ASfusion = hfusion/T.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 16% (6 reviews)
Calculations Known Heating of a cool steel bar with hot steam in a...View the full answer

Answered By

User l_1046330
I have experience of teaching since 5 years. I am also involved in content writing for the lectures. I have solved many solutions at different answer platform like Live Person, Toppr, Chegg India etc. Hence, I have an wide experience of doubt solving of various subjects like Mathematics, Science, Logical Reasoning, Advanced Mathematics etc.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
An insulated rigid tank with a volume of 7 m 3 contains steam at 500 kPa and 480 K and a hot 5-kg steel bar. The steel bar cools until the insulated system reaches an equilibrium temperature of 520...
-
A rigid tank with a volume of 1 m3 contains superheated steam at 500 kPa and 500oC. Determine (a) The mass. (b) Total internal energy (U) of the steam. The tank is now cooled until the total internal...
-
A rigid tank with a volume of 2.5 m3 contains 15 kg of saturated liquidvapor mixture of water at 75C. Now the water is slowly heated. Determine the temperature at which the liquid in the tank is...
-
A company is considering purchasing either Machine I or Machine II. The following data apply: O $21,500 O $26,575 $27,080 Parameter The annual interest rate is 10%, and all cash flows may be treated...
-
The following financial statement information is from five separate companies. Required 1. Answer the following questions about Company V: a. What is the amount of equity on December 31, 2010? b....
-
A 50.00-mL aliquot of a solution containing iron(II) and iron(III) required 10.98 mL of 0.01500 M EDTA when titrated at pH 2.0 and 23.70 mL when titrated at pH 6.0. Express the concentration of each...
-
Two rectangular plates of sizes \(L\) and \(W\) are facing each other and separated by a distance \(H\). Derive an expression for the view factor between these disks. Find the limit if \(W\) is...
-
Preston Media Corporation had the following income statement and balance sheet for 2018: Requirements 1. Compute the acquisition of plant assets for Preston Media Corporation during 2018. The...
-
3. May 9: Derby's engineering staff complete the train design and it is approved by officials from OM as well as the Transportation Safety Board. 4. August 1: Derby completes construction of the 10...
-
Apply the use of internal controls to the scenario below, then respond to the questions that follow: Ted Jones owns rental properties in Texas. Each property has a property manager, who collects the...
-
Steam in a pistoncylinder device initially has a volume of 0.2 m 3 at 300 kPa and 540 K. The steam is compressed reversibly from 300 kPa to 700 kPa at a constant temperature of 540 K. The steam...
-
An insulated rigid tank contains two chambers. Steam at 300 kPa and 500 K is contained in one chamber with a volume of 0.9 m 3 . Saturated liquid at 300 kPa is contained in the other chamber with a...
-
(a) Explain why the degree of degeneracy of an H-atom energy level is given by (b) Break this sum into two sums. Evaluate the first sum using the fact that Show that the degree of degeneracy of the...
-
Investigation into Supreme Court Justice Russell Brown's conduct under way The Canadian Judicial Council says it is reviewing a complaint into the alleged conduct of Supreme Court of Canada Justice...
-
Casey Corporation produces a special line of basketball hoops. Casey Corporation produces the hoops in batches. To manufacture a batch of the basketball hoops, Casey Corporation must set up the...
-
What governance mechanisms, regulatory frameworks, and ethical guidelines are necessary to ensure responsible innovation practices, safeguard against potential risks and unintended consequences of...
-
what ways do disruptive innovation cycles, characterized by rapid prototyping, iterative design processes, and agile development methodologies, challenge traditional notions of market stability,...
-
You have just been chosen to appear on Hoosier Millionaire! The rules are as follows: There are four hidden cards. One says STOP and the other three have dollar amounts of $150,000, $200,000, and...
-
When real estate is sold during a year, why is it necessary that the real estate taxes on the property be apportioned between the buyer and seller?
-
Assume that your audit team has established the following parameters for the examination of ELM's sales transactions: LO G-3 Risk of incorrect acceptance...
-
Replace the force system acting on the frame by an equivalent resultant force and couple moment acting at point A. A 300 N 0.5 m 30/ 1 m 500 N 0.5 m 0.3 m - - 400 N
-
The forces F1 = {-4i + 2j - 3k} kN and F2 = {3i - 4j - 2k} kN act on the end of the beam. Replace these forces by an equivalent force and couple moment acting at point O.
-
Replace the loading by an equivalent resultant force and couple moment at point O. 0.5 m y 0.7 m F =(-2i+5j-3 k) kN 0.8 m- F = [8 i-2 k} kN
-
Why is incubation process important in nurturing budding entrepreneurs?
-
explain various ways of protecting business idea
-
What are Inbound and Outbound Marketing? Explain

Study smarter with the SolutionInn App


