Determine the enthalpy change for air undergoing a process that causes a change of state from 300
Question:
Determine the enthalpy change for air undergoing a process that causes a change of state from 300 K and 1 atm to 1000 K and 5 atms. Do this in two ways: (1) Assume ideal gas behavior and use Table C.2, and (2) use the NIST software, which incorporates real gas behavior. How do the two results compare? Why do the results differ? One objective of this problem is to acquaint you with ways to find the calorific properties of air.
Table C.2
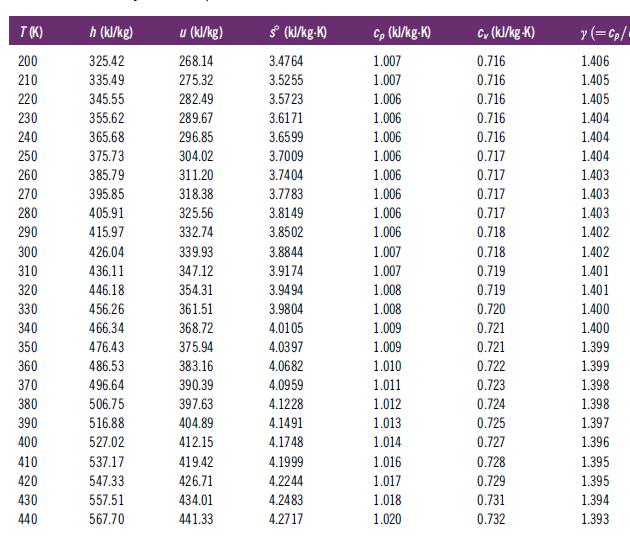
Transcribed Image Text:
T(K) 200 210 220 230 240 250 260 270 280 290 300 310 320 330 340 350 360 370 380 390 400 410 420 430 440 h (kJ/kg) 325.42 335.49 345.55 355.62 365.68 375.73 385.79 395.85 405.91 415.97 426.04 436.11 446.18 456.26 466.34 476.43 486.53 496.64 506.75 516.88 527.02 537.17 547.33 557.51 567.70 u (kJ/kg) 268.14 275.32 282.49 289.67 296.85 304.02 311.20 318.38 325.56 332.74 339.93 347.12 354.31 361.51 368.72 375.94 383.16 390.39 397.63 404.89 412.15 419.42 426.71 434.01 441.33 sº (kJ/kg-K) 3.4764 3.5255 3.5723 3.6171 3.6599 3.7009 3.7404 3.7783 3.8149 3.8502 3.8844 3.9174 3.9494 3.9804 4.0105 4.0397 4.0682 4.0959 4.1228 4.1491 4.1748 4.1999 4.2244 4.2483 4.2717 Cp (kJ/kg-K) 1.007 1.007 1.006 1.006 1.006 1.006 1.006 1.006 1.006 1.006 1.007 1.007 1.008 1.008 1.009 1.009 1.010 1.011 1.012 1.013 1.014 1.016 1.017 1.018 1.020 Cv (kJ/kg-K) 0.716 0.716 0.716 0.716 0.716 0.717 0.717 0.717 0.717 0.718 0.718 0.719 0.719 0.720 0.721 0.721 0.722 0.723 0.724 0.725 0.727 0.728 0.729 0.731 0.732 ?(=cp/ 1.406 1.405 1.405 1.404 1.404 1.404 1.403 1.403 1.403 1.402 1.402 1.401 1.401 1.400 1.400 1.399 1.399 1.398 1.398 1.397 1.396 1.395 1.395 1.394 1.393
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
I 300 K P1 atmos Given 1000 K P 5 atmos Find The change in enthalp...View the full answer

Answered By

Allan Olal
I have vast tutoring experience of more than 8 years and my primary objective as a tutor is to ensure that a student achieves their academic goals.
4.70+
78+ Reviews
412+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Use Table C.2 to calculate the mass-specific enthalpy change for air undergoing a change of state from 300 K to 1000 K. How does this value compare with that estimated using the constant-pressure...
-
Air at 300 K and 1 atm enters an in-line tube bank consisting of five rows of 10 tubes each. The tube diameter is 2.5 cm and Sn = Sp = 5.0 cm. The incoming velocity is 10 m/s and the tube wall...
-
The gas-turbine portion of a combined gas-steam power plant has a pressure ratio of 15. Air enters the compressor at 300 K and 1 atm at a rate of 13 kg/s and is heated to 1500 K in the combustion...
-
Label the following as True, False, or Uncertain and explain your choice. (Uncertain means that it can be either true or false depending upon the circumstances.) a. All members of a resource cartel...
-
Determine whether each the following statements describe variable costing (VC), full absorption costing (FA), or both (B): _____ 1. Measures gross margin as the difference between sales revenue and...
-
Are children with higher exposure to pesticides more likely to develop ADHD (attention-deficit/hyperactivity disorder)? In a recent study, authors measured levels of urinary dialkyl phosphate (DAP, a...
-
Use the summary statistics in Exercise 11 to test the null hypothesis H0: 1 = 0 versus H1: 1 0. Use the = 0.01 level of significance.
-
The Muck and Slurry merger has fallen through (see Section 33.2). But World Enterprises is determined to report earnings per share of $2.67. It therefore acquires the Wheelrim and Axle Company. You...
-
In the heating furnace, where the gas temperature is 1 4 0 0 \ deg C , the wall is built from three layers of 6 0 mm thick dinas brick, 2 5 0 mm thick red brick and 1 1 0 mm thick concrete. Air...
-
The following transactions pertain to Smith Training Company for Year 1: Jan. 30 Established the business when it acquired $45,000 cash from the issue of common stock. Feb. 1 Paid rent for office...
-
Consider an ideal gas. Indicate whether the following thermodynamic properties depend on pressure when the temperature is fixed: density, specific volume, molar-specific internal energy,...
-
In a closed (fixed-mass) system, an ideal gas undergoes a process from an initial state with pressure 517 kPa and volume of 0.1416m 3 to a final state with pressure 172.3 kPa and volume of 0.2741m 3...
-
How does the formula for the general sine function (x) = Asin ((2/B)(x - C)) + D relate to the shifting, stretching, compressing, and reflection of its graph? Give examples. Graph the general sine...
-
Marina signed a simple discount note on September 1. The discount rate on the note is 4.7%and has a maturity date of December 31. If the proceeds of the note are $12, 000, find the face value. (Use...
-
0%) Consider the sets X = [1,12] and Y = [3,18]. a) Compute XUY. b) If Z[1,20], is XcZ? Is Yc Z? Why or why not? (c) If the universe is R., then what is X and Y? 2023 Lester M. Desul. Jatinder...
-
On January 1.2022, the ledger of Sunland Company contains these liability accounts. Accounts Payable $53,500 Sales Taxes Payable 7,200 Unearned Service Revenue 16,500 During January, these selected...
-
Define the 4 x 4 matrix, A. A [3 4 4 3; 4 5 6 7; 5674; 4567] Find the content of the following matrices and check your results for content using Matlab. a) a = g(:,2) b) a = g(4,:) c) a = g(3:4, 1:3)...
-
A fluid flowing in a pipe 30cm in diameter has a uniform velocity of 4m/s. The pressure at the center of the pipe is 40KPa, and the elevation of the pipes centerline above an assumed datum is 4.5m....
-
Using the trial balance prepared in Part 3 of Problem 2-4B, prepare an income statement and statement of changes in equity for the month ended September 30, 2014, and a balance sheet at September 30,...
-
The rate at which the temperature of an object changes is proportional to the difference between its own temperature and the temperature of the surrounding medium. Express this rate as a function of...
-
Two types of plastic are suitable for use by an electronic calculator manufacturer. The breaking strength of this plastic is important. It is known that 1 , = 2 = 1.0 psi. From random samples of n...
-
The following are the burning times of chemical flares of two different formulations. The design engineers are interested in both the mean and variance of the burning times. (a) Test the hypothesis...
-
An article in Solid State Technology, "Orthogonal Design for Process Optimization and Its Application to Plasma Etching" by G. Z. Yin and D. W. Jillie (May, 1987) describes an experiment to determine...
-
You have recently become a divorced, single parent and moved to Kalamazoo, MI. You have 3 children (10, 6, 3). You were recently hired at Target- located on West Main working 11am-7pm Monday through...
-
1. Consider the following studies. College Scorecard (Institution-Level Data) Pulse Survey Beginning Postsecondary Students Longitudinal Study (BPS) International House Price Database After...
-
Have you seen this month's inflation numbers? Each time new numbers are released, the words inflation and inflation rate receive attention and trend in the news. Inflation is a general, sustained...

Study smarter with the SolutionInn App


