Determine the function: h(T) where h(T) = fcdT and C = 11.515 172 1530 0.05 + T
Question:
Determine the function: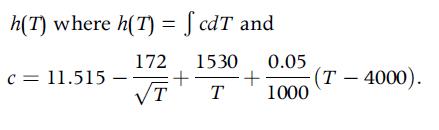
Transcribed Image Text:
h(T) where h(T) = fcdT and C = 11.515 172 1530 0.05 + T T + (T-4000). 1000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
hT fedT 11515172705 15307 ed...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Determine the voltage transfer function Vo(s)/Vc(s) as a function of s for the network shown in fig p11.3. R1 He
-
Determine the voltage transfer function Vo(s)/Vc(s) as a function of s for the network shown in fig p11.1.
-
Determine the cumulative distribution function for the distribution in Exercise 4-8. Use the cumulative distribution function to determine the probability that a length exceeds 75 millimeters....
-
Name one industry (retail, technology, biopharma, etc) that you find interesting and compelling and state why it attracts your interest. Lookup a public company in that industry - Who is it? Look at...
-
West Coast Utilities had a net profit of $900 million. It has 900 million shares outstanding and paid annual dividends of $0.90 per share. What is the dividend payout ratio?
-
Referring to Exercise 3.39 find (a) f(y\2) for all values of y; (b) P(Y = 0 | X = 2).
-
For an ideal gas (a) \(\left(\frac{\partial U}{\partial V} ight)_{T}=1\) (b) \(\left(\frac{\partial U}{\partial V} ight)_{T}=0\) (c) \(\left(\frac{\partial U}{\partial V} ight)_{T}=-1\) (d) None of...
-
Rembrandt Paint Company had the following income statement items for the year ended December 31, 2011 ($ in 000s): In addition, during the year the company completed the disposal of its plastics...
-
a) Figure 1 shows the curve y=x+4x and the x-axis. Find the area of the shaded region. y. y= x+4x Figure 1 Figure 2 2 y=6-x2 2 y=2 (5 marks) b) Figure 2 shows the shaded region bounded by the lines y...
-
A developer has purchased a laundromat and an adjacent factory. To keep smoke, which ruins the clothes, out of the dryers the developer can protect the laundromat or install filters on the factorys...
-
Determine the derivative dT/dP of the function T = 32.4P 0.5 12.8P 0.5 .
-
Do not collect any data, but use your general knowledge to draw a rough sketch of a histogram of the ages of the students at your school. Be sure to label the horizontal and vertical axes.
-
Each Revit view is saved as a separate file on your hard drive. A) True B) False
-
You cannot adjust the depth of view using the crop window. A) True B) False
-
Fill in the blank field in this text: With the elevation tag selected, you can use the [1]___________________________________ icon to adjust the tag orientation to look at an angled wall.
-
You cannot adjust the extent of view using the crop region. A) True B) False
-
You can use any Windows True Type font in Revit. A) True B) False
-
Calculate the pH of a solution made up from 2.0 g of potassium hydroxide dissolved in 115 mL of 0.19 M perchloric acid. Assume the change in volume due to adding potassium hydroxide is negligible.
-
A company pledges their receivables so they may Multiple Choice Charge a factoring fee. Increase sales. Recognize a sale. Collect a pledge fee. Borrow money. Failure by a promissory notes' maker to...
-
Why do we say that the equilibrium constant for the reaction H 2 O H + + OH - (or any other reaction) is dimensionless?
-
Write the expression for the equilibrium constant for each of the following reactions. Write the pressure of a gaseous molecule, X, as P X . a. b. 3Ag*(aq) + PO (aq) = Ag3PO4(s)
-
(a) A favorable entropy change occurs when S is positive. Does the order of the system increase or decrease when S is positive? (b) A favorable enthalpy change occurs when H is negative. Does the...
-
Solve the Linear Systems by the method of elimination 3x+4y-z = 8 6x+8y2z = 3
-
The spam data file contains 4601 emails, 1813 of which are spam. The file has 57 features that include indicators for the presence of 54 keywords (e.g. free, deal, ! etc), counts for capitalized...
-
3 11-3 Consider the given matrix A = 06-2 5 2 1 (i) Compute the det(A) by using the cofactor expansion method. (ii) Determine the adj(A). (iii) Determine the inverse, A.

Study smarter with the SolutionInn App


