For the paths indicated in the sketch, show that the entropy change for an ideal gas by
Question:
For the paths indicated in the sketch, show that the entropy change for an ideal gas by either path is the same.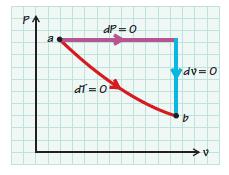
Transcribed Image Text:
PA dP=0 dl=0° dv=0 b A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The entropy change of an ideal gas undergoing a reversible process can be calculated using the equat...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Using the equation for the entropy change of an ideal gas when the volume and temperature change and TV 1 is a constant, show explicitly that the entropy change is zero for a quasi-static adiabatic...
-
Obtain the relation between the molar entropy change of an ideal gas vapor mixture and the molar entropy of each constituent.
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
If accounting-based performance measures, such as net income or core earnings, are to be significant components of manager compensation plans, they need to be sensitive and precise measures of...
-
Use the information in Exercise to prepare an October statement of retained earnings for Real Answers. In Exercise, On October 1, Keisha King organized Real Answers, a new consulting firm. On October...
-
Treatment of hydroxylamine (H2NOH) with an excess of Fe(III) results in the formation of N2O and an equivalent amount of Fe(II): 2H2NOH + 4Fe3+ ( N2O(g) + 4Fe2+ +4H+ + H2O
-
Find the temperature at which the surface tension of water becomes zero. What is the physical significance of this temperature?
-
A successful California engineer has installed a circular hot tub in his back yard and finds that for the typical operating conditions prescribed below, water must be added at a rate of 0.00 I kg/s...
-
Each item below describes an amount(s) not reflected in the financial statements of Drake Company. Select from the option list provided the amount, if any, required to be recognized in Drake's...
-
Suppose the following table shows the number of labor hours needed to produce airplanes and automobiles in the United States and South Korea, but one of the numbers is unknown. a. Without knowing the...
-
For a simple compressible substance with c p = a(1 + bT), where a and b are constants, determine the entropy change for an isobaric process going from T 1 and T 2 .
-
Air expands through an air turbine from inlet conditions of 690 kPa and 538 C to an exit pressure of 6.9 kPa in an isentropic process. Determine the inlet specific volume, the outlet specific volume,...
-
What are some other areas of the current food label that are misleading?
-
On December 15, 2020 Chan Barry purchased a new camper (to be pulled behind his truck) from Manson Building and Spas, Inc. (Manson) for $37,219. Barry testified that he purchased the camper so that...
-
Section One: Quotations . Instructions: In a word document. For FOUR of the quotations below, you are to 1) indicate which number quotation you are discussing, 2) name the philosophy/philosopher from...
-
EXERCISE 1 This exercise is about offer and acceptance. John, a homeowner, answers the telephone and listens to a solicitor make a five-minute sales pitch for Weed Gardening Services. John responds,...
-
When choosing to enable Multiple Inventory Sites tracking, what specific concern needs to be considered?
-
How are accrued real estate taxes that have been assessed but not yet due handled in a real estate transaction?
-
Assume all of the same facts as in Problem I:6-55, except that during the year Kim rents the condominium a total of 14 days. How does Kim report the income and deductions from the property?
-
When a company has a contract involving multiple performance obligations, how must the company recognize revenue?
-
Members AB and BC can each support a maximum compressive force of 800 lb, and members AD, DC, and BD can support a maximum tensile force of 2000 lb. If a = 6 ft, determine the greatest load P the...
-
Determine the force in each member of the truss and state if the members are in tension or compression. Set P1 = 10 kN, P2 = 8 kN. F E 2 m D. A B 2 m P2
-
Determine the force in each member of the truss and state if the members are in tension or compression. Set P 1 = 8 kN, P 2 = 12 kN. F E 2 m D. A B 2 m P2
-
For the data set given find the output for the new instance = (Red, Suv, Domestic) www Sr Colour Type Origin Stolen wwwwwwwww 1 RED SPORTS DOMASTIC YES 2 RED SPORTS DOMASTIC NO 3 RED SPORTS DOMASTIC...
-
What are the postfix and prefix forms of the expression? A+B*(C-D)/(P-R).
-
Write short note function prototype, accessing function and utility function. Explain Features of the Object Oriented programming.

Study smarter with the SolutionInn App


