How to Interpolate. Apply interpolation to the property data in Table B.1 to determine the following properties
Question:
How to Interpolate. Apply interpolation to the property data in Table B.1 to determine the following properties of saturated liquid H2O:A. The specific volume at 300.7 KB. The saturation pressure at 282.6 KC. The mass-specific enthalpy at 514 K
Table B.1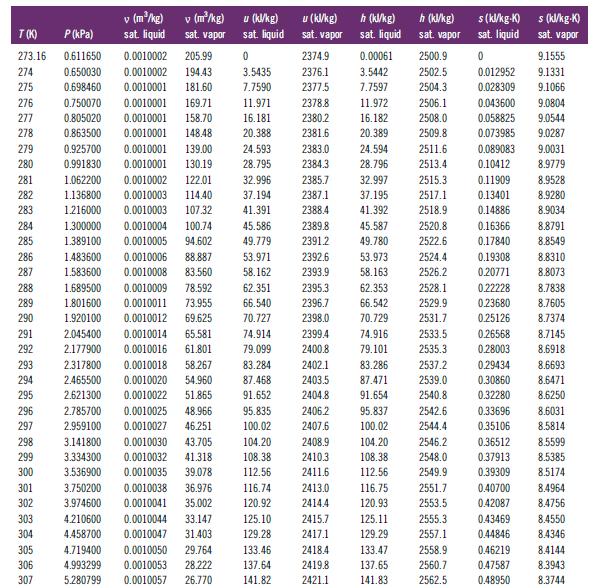
Transcribed Image Text:
276 277 278 279 280 281 282 283 284 285 286 287 T(K) P (kPa) 273.16 0.611650 274 0.650030 275 0.698460 0.750070 0.0010001 169.71 0.805020 0.0010001 158.70 0.863500 0.0010001 148.48 0.925700 0.0010001 139.00 24.593 0.991830 0.0010001 130.19 28.795 1.062200 0.0010002 122.01 32.996 2385.7 1.136800 0.0010003 114.40 37.194 2387.1 41.391 2388.4 1.216000 0.0010003 107.32 1.300000 0.0010004 100.74 45.586 2389.8 1.389100 0.0010005 94.602 49.779 2391.2 0.0010006 88.887 53.971 0.0010008 83.560 58.162 288 289 290 296 297 298 299 300 1.483600 1.583600 301 302 303 304 305 306 307 1.689500 0.0010009 78.592 1.801600 0.0010011 1.920100 2.045400 291 292 2.177900 293 2.317800 0.0010018 58.267 294 2.465500 0.0010020 54.960 295 2.621300 51.865 0.0010022 0.0010025 48.966 2.785700 2.959100 0.0010027 46.251 3.141800 0.0010030 43.705 3.334300 0.0010032 41.318 3.536900 0.0010035 39.078 0.0010038 36.976 0.0010041 35.002 V (m³/kg) sat. liquid 3.750200 3.974600 v (m²/kg) sat. vapor 4.210600 4.458700 0.0010002 205.99 0.0010002 194.43 0.0010001 181.60 u (kW/kg) sat. liquid 0 3.5435 7.7590 62.351 66.540 73.955 0.0010012 69.625 70.727 0.0010044 33.147 0.0010047 31.403 11.971 16.181 20.388 0.0010014 65.581 74.914 0.0010016 61.801 79.099 83.284 87.468 91.652 95.835 100.02 104.20 108.38 112.56 4.719400 0.0010050 29.764 4.993299 0.0010053 28.222 5.280799 0.0010057 26.770 116.74 120.92 125.10 129.28 u (kJ/kg) sat. vapor 133.46 137.64 141.82 2374.9 2376.1 2377.5 2378.8 2380.2 2381.6 2383.0 2384.3 2392.6 2393.9 2395.3 2396.7 2398.0 2399.4 2400.8 2402.1 2403.5 2404.8 h (kJ/kg) sat. liquid 2413.0 2414.4 2415.7 2417.1 2418.4 2419.8 2421.1 0.00061 3.5442 7.7597 11.972 16.182 20.389 45.587 49.780 2506.1 2508.0 2509.8 24.594 2511.6 28.796 2513.4 32.997 2515.3 37.195 2517.1 41.392 2518.9 2528.1 2529.9 2531.7 2533.5 2535.3 2537.2 2539.0 2540.8 2406.2 95.837 2542.6 2407.6 100.02 2544.4 2408.9 2546.2 2410.3 2548.0 2411.6 83.286 87.471 91.654 h (kJ/kg) sat. vapor 104.20 108.38 112.56 2500.9 2502.5 2504.3 116.75 120.93 125.11 129.29 133.47 137.65 141.83 2520.8 2522.6 0.16366 0.17840 53.973 2524.4 0.19308 58.163 2526.2 0.20771 62.353 66.542 70.729 74.916 79.101 2549.9 2551.7 2553.5 2555.3 2557.1 s(kl/kg-K) sat. liquid 0 2558.9 2560.7 2562.5 0.012952 0.028309 0.043600 0.058825 0.073985 0.089083 0.10412 0.11909 0.13401 0.14886 0.22228 0.23680 0.25126 0.26568 0.28003 0.29434 0.30860 0.32280 0.33696 0.35106 0.36512 0.37913 0.39309 0.40700 0.42087 0.43469 0.44846 0.46219 0.47587 0.48950 s (kJ/kg-K) sat. vapor 9.1555 9.1331 9.1066 9.0804 9.0544 9.0287 9.0031 8.9779 8.9528 8.9280 8.9034 8.8791 8.8549 8.8310 8.8073 8.7838 8.7605 8.7374 8.7145 8.6918 8.6693 8.6471 8.6250 8.6031 8.5814 8.5599 8.5385 8.5174 8.4964 8.4756 8.4550 8.4346 8.4144 8.3943 8.3744
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Apply interpolation to find the following properties of saturated liquid water from Table B1 ...View the full answer

Answered By

AJIN kuriakose
I have completed B.Tech in Electrical Engineering & Masters in Power & Control From one of the best universities in India. I got the 99.05 percentile in the Gate Electrical Engineering Exam. I can Help students solving assignments in Electrical subjects like Power Electronics, Control system, Analog, Network Theory & Engineering Mathematics. Clear your fundamentals and develop problem-solving skills and analytical skills to crack the exam.
Get guidance and the opportunity to learn from experienced...
I can provide tuition for Electrical engineering subjects (Power Electronics, Digital electronics, Network Theory, Control System & Engineering Mathematics). The toughest subject of Electrical engineering can be made simple in online classes...
I can also solve it.
1 .I can help you with your assignments or exams or quiz or tutoring.
2. Very strict to the deadlines.
Message me for any help in assignments, live sessions. I am here to help students for all assignments, tests and exams and I will make sure you always get _95% In your subject.
Contact me in solution inn for any help in your semester, projects and for many more things . Also feel free to contact me through solution inn and for any advise related to tutoring and how it works here.thank you.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
How to Interpolate. Apply interpolation to the property data in Table B.2 to determine the following properties of saturated vapor H 2 O:A. The specific volume at 50.8 kPa.B. The saturation...
-
Apply interpolation to the property data in Table B.3 to determine the following properties of superheated steam: A. The specific volume at 523 K and 1.0 MPa B. The mass-specific enthalpy at 815 K...
-
Apply interpolation to the property data in Table B.3 to determine the following properties of superheated steam: A. The specific volume at 727 K and 1.5 MPaB. The mass-specific enthalpy at 823 K and...
-
What are the Marketing Cost Estimates of Pepsi Company? Marketing estimates, in 2013-2019? It can be write in a paragraph and explain it statistically.
-
You have been asked to analyze the capital structure of DASA, an environmental waste disposal firm, and make recommendations on a future course of action. DASA has 40 million shares outstanding,...
-
1. Unless we reduce the incidence of child abuse, future crime rates will increase. (R, F) 2. If pharmaceutical makers conceal test results, they are subject to substantial fines. (P, S) 3. African...
-
Sunny Day Sunlamps, Inc., had Sales Revenue of $53 million, Sales Returns and Allowances of $2 million, and Sales Discounts of $0.3 million in 2010. Cost of goods sold was $23 million, and net income...
-
A Carnot cycle uses 1.00 mol of a monatomic perfect gas as the working substance from an initial state of 10.0 atm and 600 K. It expands isothermally to a pressure of 1.00 atm (Step 1), and then...
-
Today, the spot price of the EUR/USD exchange rate is $1.0796. The bid and ask quotes for the nine-month EUR/USD forward contracts are, respectively, 73.56 and 75.68. According to the interest rate...
-
Recall the following details about the two employees hired in November. a. CPP is deducted at 5.10% (considering $3,500 yearly exemption). b. EI is deducted at 1.62% of gross earnings. c. Both...
-
Steam expands is entropically (i.e., at constant entropy s) from 2MPa and 500 K to a final state in which the quality is 0.90. Determine the final-state temperature, pressure, and specific volume. T...
-
In a proposed automotive steam engine, the steam after expansion would reach a state at which the pressure is 200 kPa (gage) and the specific volume is 0.2997 m 3 /kg. Atmospheric pressure is 100...
-
A helicopter 120 m from a person takes off vertically. Express the distance d from the person to the helicopter as a function of the height h of the helicopter. What are the domain and the range of d...
-
In 2019, Lori Jerin leased the mineral rights in a property in Wyoming, agreeing to a 25% royalty. The joint working interest in the property is owned by the following companies: In 2020, a...
-
After deciding to use the full cost method of accounting, Green Petroleum began operations on January 1, 2018. Transactions for the first three years include the data below. 2019 1) A delay rental of...
-
Lamar Petroleum started its oil and gas exploration and production business by acquiring leases in both the United States and Canada. Data for 2018 and 2019 follow: REQUIRED: a. Record the above...
-
Fitzgerald Corporation began operating on 1/1/18. At 12/31/18, the company owned the following leases in the United States: The production was sold at $150/bbl and $5.50/Mcf. Current prices at...
-
Abbot Oil Corporation, located in Houston, Texas, has been operating for four years. Abbot uses full cost accounting and excludes all possible costs from the amortization base. The following account...
-
Is a retailer's desire to build up its private-label brands an inevitable source of channel conflict? Explain your answer.
-
Multiple Choice Questions: 1. The largest component of aggregate demand is? a. Government purchases. b. Net exports. c. Consumption. d. Investment. 2. A reduction in personal income taxes, other...
-
A brick wall 3 m high, 7.5 m wide, and 200 mm thick has a thermal conductivity of 0.7 W/(m C). The temperature on the inner face is 25C, and the temperature on the outer face is 0C. How much heat is...
-
A 2500-lb automobile comes to a complete stop from 65 mph. If 60% of the braking capacity is provided by the front disk brake rotors, determine their temperature rise. Each of the two cast-iron...
-
A small hydroelectric power plant operates with 500 gal of water passing through the system each second. The water falls through a vertical distance of 150 ft from a reservoir to the turbines....
-
Write, compile, and test the MovieQuote Info class so that it displays your favorite movie quote, the movie it comes from, the character who said it, and the year of the movie. An example of the...
-
we have seen how we can use an implication of Central Limit Theorem to obtain a statistically significant mean running time measurement of an implementation of an algorithm. Let's say we have N = 25...
-
Officer Jablonski was on routine patrol when she observed a 2002 Blue Jeep Liberty not using the turn signal when switching lanes and swerving between lanes not staying within the lines. After...

Study smarter with the SolutionInn App


