Methane stored in a tank at 8 MPa and 300 K is used as a fuel for
Question:
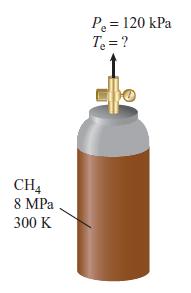
Methane stored in a tank at 8 MPa and 300 K is used as a fuel for a laboratory-scale gas-turbine combustor. The methane is throttled as it passes through a pressure regulator. The regulated pressure is 120 kPa. Determine the temperature of the methane at the exit of the pressure regulator. Assume the process is adiabatic and neglects any kinetic energy changes.
Transcribed Image Text:
CH4 8 MPa 300 K P₂ = 120 kPa T₂ = ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To solve this problem we can use the adiabatic throttling equation which rela...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Methane at 10 MPa and 300 K is heated at constant pressure until its volume has increased by 80 percent. Determine the final temperature using the ideal gas equation of state and the compressibility...
-
The vortex tube (also known as a Ranque or Hirsch tube) is a device that produces a refrigeration effect by expanding pressurized gas such as air in a tube (instead of a turbine as in the reversed...
-
Methane gas is stored in a tank at a pressure of 15 bar and 294 K. The tank has a capacity of 0.057 m^3. Gas is allowed to escape from the tank through a partially opened valve. When the pressure in...
-
Assume that everything is the same for the project except the marginal tax rate is now 35%. What is the new NPV? Assume that the cost per unit can be decreased to $12 by using cheaper raw materials...
-
Use the information in the adjusted trial balance reported in Exercise to prepare Resource Trucking Companys classified balance sheet as of December 31, 2011. In Exercise Account Title Debit Credit...
-
If you invest $8,000 per period for the following number of periods, how much would you have? a. 10 years at 5 percent b. 20 years at 9 percent c. 35 periods at 11 percent
-
PWC Corp. accounting records include the following items, listed in no particular order, at December 31, 2008 : Prepare PWC's income statement for the year ended December 31, 2008. Omit earnings per...
-
Mitachlordion Technology, Inc. (MTI), has two divisions: Birmingham and Tampa. Birmingham currently sells a diode reducer to manufacturers of aircraft navigation systems for $1,550 per unit. Variable...
-
Wildhorse Co. reports the following information (in millions) during a recent year: net sales, $11,750.0; net earnings, $587.5; total assets, ending, $5,200.0; and total assets, beginning, $4,200.0....
-
Samuel Cox, owner of Cox Video Center, sent the income statement shown on the next page to several of his creditors who had asked for financial statements. The business is a sole proprietorship that...
-
Sketch an isentropic and actual pumping process on a Ts diagram for a compressed liquid. Include the vapor dome and the pressure contours that pass through the initial and final states.
-
In a refrigerator, saturated liquid R-134a (a refrigerant) is throttled from an initial temperature of 305 K to a final pressure of 80 kPa. Determine the final temperature and specific volume of the...
-
Analyze BPs efforts to improve sustainability. Do you think they are sufficient, or does the company need to do more? BP, formerly British Petroleum and the Anglo-Persian Oil Company, has experienced...
-
For the following substances, give the electron pair geometry, molecular geometry, and hybridization of the central atom(s). Put your answer in the following format; electron pair geometry, molecular...
-
c. Find the PV of $1,000 due in 6 years if the discount rate is 8%. Round your answer to the nearest cent. $ d. A security has a cost of $1,000 and will return $3,000 after 6 years. What rate of...
-
Your company has bonds that mature in 10 years and have a face value of $1,000. The bonds have an 8 percent quarterly coupon (that is, the nominal coupon rate is 8 percent and hence pays $20 every...
-
Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise inventory Prepaid expenses Plant assets, net Total assets Liabilities and Equity...
-
Lisa is a graduate student from Holmes Institute who is actively involved in investment in the securities market. She had established one investment portfolio 5 years ago. Required: Lisa's portfolio...
-
Required For each of the following controls: (a) Identify whether it is preventive or detective, and (b) Indicate whether it relates to the control objective of authorization. 1. The computerized...
-
What recommendations would you make to Big Four firms to help them (1) avoid confrontations with governmental officials in an authoritarian society and (2) deal effectively with such confrontations...
-
Represent each of the following as a number between 0.1 and 1000 using an appropriate prefix: (a) 45320 kN, (b) 568(105) mm, (c) 0.00563 mg.
-
Round off the following numbers to three significant figures: (a) 58.342 m, (b) 68.534 s, (c) 2553 N, (d) 7555 kg.
-
Represent each of the following combinations of units in the correct SI form using an appropriate prefix: (a) Mg/mm, (b) mN/s, (c) mMg
-
Zerox Copying Company plans to borrow $225,000. New Jersey National Bank will lend the money at one-half percentage point over the prime rate at the time of 16.50 percent (17 percent total) and...
-
Harper Engine Company needs $623,000 to take a cash discount of 2.50/10, net 85. A banker will lend the money for 75 days at an interest cost of $18,300. a. What is the effective rate on the bank...
-
Several factors may influence interest rates for individual securities in an economy like the United Kingdom. One such factor is inflation. Explain how the factor may influence the interest rates of...

Study smarter with the SolutionInn App


