One mole of a hydrocarbon fuel (CH x ) is burned with excess air. The volumetric analysis
Question:
One mole of a hydrocarbon fuel (CHx) is burned with excess air. The volumetric analysis of the dry products (with H2O removed) yields: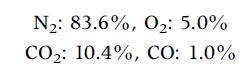
A. Determine the approximate composition of the fuel on a mass basis.B. Determine the percent theoretical air.C. Determine the equivalence ratio.
Transcribed Image Text:
N₂: 83.6%, O₂: 5.0% CO₂: 10.4%, CO: 1.0%
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
A To determine the approximate composition of the fuel on a mass basis we need to do a mass balance on the carbon and hydrogen in the fuel Since we do...View the full answer

Answered By

Diana Muriuki
As an online math tutor, I have several years of hands-on experience working with students of all ages and skill levels. I hold a Bachelor's degree in Mathematics and a Master's degree in Education. Additionally, I have completed multiple training courses in online teaching and tutoring methods.
Throughout my career, I have worked with students in both individual and group settings, including classroom teaching, after-school tutoring, and online instruction. I am proficient in teaching a wide range of math topics, from basic arithmetic to advanced calculus and statistics.
One of my greatest strengths as a tutor is my ability to adapt my teaching style to meet the unique needs and learning styles of each individual student. I understand that every student is different, and I strive to create a comfortable and supportive learning environment that encourages growth and development.
In addition to my formal education and tutoring experience, I am also a lifelong learner with a passion for mathematics. I am constantly seeking out new resources and methods to improve my own knowledge and skills, and I believe this passion and enthusiasm helps to inspire my students as well.
Overall, my hands-on experience and proficiency as a math tutor are grounded in a combination of formal education, practical experience, and a genuine love of mathematics. I am confident in my ability to help students achieve their goals and succeed in math, and I look forward to the opportunity to work with new students and continue to grow as an educator.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
The combustion of a hydrocarbon fuel with air results in a mixture of products of combustion having the composition on a volume basis as follows: 4.89 percent carbon dioxide, 6.50 percent water...
-
A hydrocarbon fuel CaHb is burned with dry air. Determine the composition of the fuel combusted (whole number atoms) and the percent theoretical air if a dry volumetric analysis of the products...
-
n-Pentane is burned with excess air in a continuous combustion chamber. (a) A technician runs an analysis and reports that the product gas contains 0.270 mole% pentane, 5.3% oxygen, 9.1% carbon...
-
Consider a set of documents. Assume that all documents have been normalized to have unit length of 1. What is the "shape" of a cluster that consists of all documents whose cosine similarity to a...
-
Most airlines in the United States have frequent flier programs that grant free flights if a customer accumulates enough flight miles on the airline. For example, United Airlines offers a free...
-
Discuss the advantages that result from the clientserver architecture of the X Window system.
-
Explain the purpose of a business and describe how businesses are organised and structured.
-
During the current year, Ross Corporation purchased a warehouse for $45,000, paid $1,000 dividends, sold land for $7,900, and issued $25,000 of common stock. All of these were cash transactions. Show...
-
In the epicyclic gear train shown in Fig. 15.36, the compound wheels E and F rotate freely on shaft A which carries the planet carrier G. The planets B and C are compounded gears. The number of teeth...
-
To: [Your name] From: Supervising Attorney Re: Eldridge v. Eldridge; modification of child support We represent Gwen Eldridge in the case of Eldridge v. Eldridge. The Eldridgeswere divorced in 2010....
-
Ethane burns with 150% stoichiometric air. Assume the air is 79% N 2 and 21%O 2 by volume. Combustion goes to completion. Determine (a) The airfuel ratio by mass (b) The mole fraction (percentage) of...
-
Natural gas is burned to produce hot water to heat a clothing store. Assuming that the natural gas can be approximated as methane (CH 4 ) and that the air is a simple mixture of O 2 and N 2 in molar...
-
Two point charges are located on the x -axis: a charge of + 6.0 nC at x = 0 and an unknown charge q at x = 0.50 m. No other charges are nearby. If the electric field is zero at the point x = 1.0 m,...
-
1. Find the regular expression for the following finite automaton using ardens theorem? a F
-
what ways does the loss of genetic diversity within a population impact its adaptive potential, and how might this contribute to the overall vulnerability of a species to environmental changes ?
-
Gather than focus on the treatment of chronic disease, policies that influence population health tend to emphasize prevention and wellness; the reduction or elimination of waste, and the eradication...
-
According to the inheritance hierarchy in the following Figure 1, and the provided Shape class and Circle class: Shape - area (double) -color (String) - name(String) + Shape(String, String) +...
-
2. Draw the graph represented by the adjacency matrix with vertices a, b, c, d, e, f A = 3. Draw the graph represented by the incidence matrix with vertices: V1, V2, V3, V4, V5, 4 1 1 1 0 2 1 0 1 1 1...
-
Circle and name the functional group in each compound. a. b. c. d. CH3-C CH2CH2CH3 HO-C-CH2CH3 H-C CH2CH3
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Invoking the usual normality assumptions, find an expression for the probability that a negative estimate of a variance component will be obtained by the analysis of variance method. Using this...
-
Analyze the data in Problem 12-9, assuming that operators are fixed, using both the unrestricted and the restricted form of the mixed models. Compare the results obtained from the two models. Problem...
-
Consider the variance components in the random model from Problem 12-9. (a) Find an exact 95 percent confidence interval on 2 . (b) Find approximate 95 percent confidence intervals on the other...
-
A company had $100,000 in short-term debt at December 31, 2019. The company refinanced this liability on a long term basis subsequent to the end of the year but before the financial statements were...
-
Kanban Company estimated sales of 40,000 units at $6 each. Budgeted cost of goods sold per unit includes $1.20 of direct materials, six minutes of direct labor time at $15 per hour, and unit overhead...
-
Wonderland Ltd. produces and sells strategy board games to game shops and toy stores. Its marketing agent has been approached by the organizers of a gaming convention who want to purchase 40,000...

Study smarter with the SolutionInn App


