Show that Eq. 12.25 can be expressed equivalently on (a) A per unit mass of mixture basis
Question:
Show that Eq. 12.25 can be expressed equivalently on
(a) A per unit mass of mixture basis
(b) A per unit mass of fuel basis.
Eq. 12.25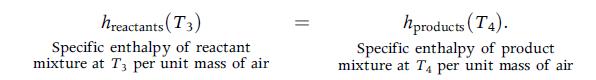
Transcribed Image Text:
hreactants (T3) Specific enthalpy of reactant mixture at T3 per unit mass of air hproducts (T4). Specific enthalpy of product mixture at T4 per unit mass of air
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
To show that Eq 1225 can be expressed equivalently on a per unit mass of mixture basis and a per unit mass of fuel basis we need to rewrite the equati...View the full answer

Answered By

Diana Muriuki
As an online math tutor, I have several years of hands-on experience working with students of all ages and skill levels. I hold a Bachelor's degree in Mathematics and a Master's degree in Education. Additionally, I have completed multiple training courses in online teaching and tutoring methods.
Throughout my career, I have worked with students in both individual and group settings, including classroom teaching, after-school tutoring, and online instruction. I am proficient in teaching a wide range of math topics, from basic arithmetic to advanced calculus and statistics.
One of my greatest strengths as a tutor is my ability to adapt my teaching style to meet the unique needs and learning styles of each individual student. I understand that every student is different, and I strive to create a comfortable and supportive learning environment that encourages growth and development.
In addition to my formal education and tutoring experience, I am also a lifelong learner with a passion for mathematics. I am constantly seeking out new resources and methods to improve my own knowledge and skills, and I believe this passion and enthusiasm helps to inspire my students as well.
Overall, my hands-on experience and proficiency as a math tutor are grounded in a combination of formal education, practical experience, and a genuine love of mathematics. I am confident in my ability to help students achieve their goals and succeed in math, and I look forward to the opportunity to work with new students and continue to grow as an educator.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
A steady stream of equimolar N2 and CO2 mixture at 100 kPa and 18C is to be separated into N2 and CO2 gases at 100 kPa and 18C. Determine the minimum work required per unit mass of mixture to...
-
A mixture that is 15 percent carbon dioxide, 5 percent carbon monoxide, 10 percent oxygen, and 70 percent nitrogen by volume undergoes an adiabatic compression process having a compression ratio of...
-
Shreya commutes in a CNG fitted van to school every day along with many other students. She told the van driver to get the CNG connection certified and timely checked it for any leakage or loose...
-
Disposal Services, Inc., a waste management company, had 3.5 million shares of common stock authorized on August 31, 20X2. Shares issued were 2 million. There were 375,000 shares held in the...
-
Bayside Coatings Company purchased waterproofing equipment on January 2, 2011, for $190,000. The equipment was expected to have a useful life of four years and a residual value of $9,000. Instruction...
-
The fuel cell (a) Produces power at low cost at considerable periods (b) Requires quite extensive maintenance (c) Has harmful by-products (d) None of these.
-
The return on investment measure may be biased in favor of divisions with older plant and equipment. Explain.
-
The US Dollar's reserve currency status confers many advantages. How did it aid the US in responding to the Russian invasion of Ukraine? What are some of the consequences of these actions and do they...
-
The A. J. Swim Team soon will have an important swim meet with the G. N. Swim Team. Each team has a star swimmer (John and Mark, respectively) who can swim very well in the 100- yard butterfly,...
-
Consider a stoichiometric reaction involving liquid octane and oxygen in a steady low reactor. The reactants enter at 25 C and 1 atm, and the products exit at the same conditions. Assuming liquid...
-
Anticipating the coming of the hydrogen economy, an enthusiastic mechanical engineer invents a steam generator. Pure hydrogen, H 2 , enters the wellinsulated combustion chamber at 298 K with a flow...
-
The oxide MnO 2 reacts with limited amounts of carbon,C(s), to produce Mn(s) and CO(g). Write the balanced equation, and calculate the amount of carbon needed to produce 22.8 L CO(g) at 1.00 atm and...
-
If you were about to receive a 30 year fixed interest mortgage for $280,000 at 3.3% interest, what would your monthly payments be?
-
The following are summary financial statements for Kuala Lumpur DIY Plc. Non-current assets Current assets Total assets 20X4 $000 20X9 $000 115 410 650 1,000 765 1,410 Capital and reserves 210 270...
-
Suppose the expected return on the market is 14%, market volatility is 27%, and the risk-free rate is 2%. a. Suppose stock A has a beta of 1.25. According to the CAPM, what is its expected return?...
-
A startup has 1,000 shares outstanding. A VC offers to invest $300,000 at a pre-money price of $1,500 per share. What is the pre-money valuation? Give your answer in millions of dollars (for example,...
-
A startup has 1,000 shares outstanding. A VC offers to invest $500,000 at a pre-money valuation of $800,000. What is the post-money valuation? Give your answer in millions of dollars (for example, if...
-
Marta borrowed $1750 from Jasper on November 15, 2008, and agreed to repay the debt with simple interest at the rate of 7.4% on June 3, 2009. How much interest was owed on June 3?
-
Which of the followingcarbocations is the least stable? CH3CH2 . CH3CHCH3 CH3 I . CH3C0 T CH3 IV. V. CH3 CH3CCH2 CH3
-
Outline the analysis of variance table for the 34 design in nine blocks. Is this a practical design?
-
Consider the data in Problem 9-3. If ABC is confounded in replicate I and ABC 2 is confounded in replicate II, perform the analysis of variance. Problem 9-3. An experiment was performed to study the...
-
Consider the data from replicate I of Problem 9-3. Suppose that only a one-third fraction of this design with I = ABC is run. Construct the design, determine the alias structure, and analyze the...
-
1. The figure provided shows a section of a very long rod of radius a = 1.68 mm and linear charge density 3.04 nC/m. The rod is inside a coaxial cylindrical shell of radius b = 11.5 mm and linear...
-
1. Two charges are set on the x -axis 9.90 cm away from each other. The charges are -8.40 nC and 18.3 nC. Calculate the electric potential at the point on the x-axis where the electric field due to...
-
Find the magnetic field (in milli-teslas, mT) that will produce a deflection of 0.30 radians for an electron beam in Thomson's experiment. The distance between the plates is 2.0 cm, the potential...

Study smarter with the SolutionInn App


