Water at 10.0 MPa (absolute) is heated from 350 K to 380 K. Determine the change in
Question:
Water at 10.0 MPa (absolute) is heated from 350 K to 380 K. Determine the change in mass-specific enthalpy for this process in three ways.
(1) Use the tables in Appendix B, interpolating as required.
(2) Use the NIST software or online database.
(3) Use the liquid property approximation, Eqn. 2.41c. Compare your results and discuss.
Eqn. 2.41c.
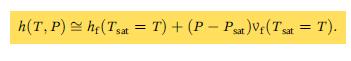
Transcribed Image Text:
h(T, P) he(Tsat = T) + (P - Psat)Vt (Tsat = T).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
We will solve the problem in three ways as instructed Solution 1 Using Tables in Appendix B From the ...View the full answer

Answered By

Aruna Mohandas
I have experience in tutoring students on various aspects of communication, such as how to structure and deliver effective presentations, how to improve their writing skills, and how to communicate effectively in a professional setting.
I am skilled in providing personalized feedback and guidance to help students improve their communication skills. I can analyze their communication styles and identify areas where they can improve, and provide practical strategies and exercises to help them achieve their goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Water at 200 kPa-gage is heated from 17 C to 46 C. The atmospheric pressure is 100 kPa. Determine the change in mass specific enthalpy for this process.
-
Compare the specific enthalpy change calculated in Problem 2.69 with the change determined directly from the ideal-gas tables. Also compare these values with that obtained from the NIST software or...
-
Determine the change in Enthalpy (h) and Entropy (s) of nitrogen as it undergoes a change of state from 200 K and 6 MPa to 300 K and 10 MPa by treating nitrogen as a perfect gas. What-if Scenario:...
-
Glass bottles can be either recycled (crushed and re-melted) or reused. The market will tend to choose the cheapest path. What factors will tend to affect the relative cost of these options? Is the...
-
Refer to the Tan-O-Rama data in M5-7. Suppose Sherri performed a least-squares regression and obtained the following results: Coefficients Intercept .....5,046.12 x Variable 1 .... 2.19 Put Sherris...
-
Venkat and Company manufactures rotators for automobile engines: R1 and R2. Based on a plantwide overhead rate, the cost estimates for two of the firms models for the current year are as follows:...
-
The cantilever beam is subjected to a couple moment \(M_{0}\) applied at its end. Determine the slope of the beam at \(B\). EI is constant. -L- B Mo
-
On October 31, 2015, Damon Companys general ledger shows a checking account balance of $8,397. The companys cash receipts for the month total $74,320, of which $71,295 has been deposited in the bank....
-
Company XYZ is a wholesale club chain store, such as Costco or Sam's Club. You, as a data analyst, are asked to perform the following data clean up, data transformation, and data calculation tasks in...
-
1. How did Joe Shaughnessy seem to do in achieving the four bottom lines? 2. Did Joe balance with the four bottom lines at the same time, or did some come before others? 3. How did Joe use the...
-
How to Define a Thermodynamic State. Given the following property data for H 2 O, designate the region, line, or point in Tv or P v space (i.e., compressed liquid, liquidvapor mixture, superheated...
-
Consider an ideal gas. Indicate whether the following thermodynamic properties depend on pressure when the temperature is fixed: density, specific volume, molar-specific internal energy,...
-
Spherical refracting surfaces an object O stands on the central axis of a spherical refracting surface. For this situation, each problem in Table 34-5 refers to the index of refraction n1 where the...
-
outstanding stock is composed of 1 0 , 0 0 0 shares of $ 1 0 0 par, noncumulative, preferred 8 % stock, and 5 0 , 0 0 0 shares of no - par common stock. Preferred dividends have been paid every year...
-
Draw a flowchart to find if the user's input number is a prime number. Assume the number is an integer.
-
es River Signorini works for New & Old Apparel, which pays employees on a semimonthly basis. River's annual salary is $174,500. Required: Calculate the following: Note: Round your final answers to 2...
-
Suppose that a spherical vapor bubble of radius R(t) is growing in a supersaturated liquid. Assume that the gas density (PG) and liquid density (PL) are each constant, and let the origin correspond...
-
Gama Ltd. has to select a capital project between Project A and Project B. The details of both the projects are given below: Cash Flows Year Project A Project B O -2000 -2000 1 1500 150 2 700 850 3...
-
You have just received a letter from a customer who purchased new rims for his car from your tire store several months ago. He paid for the rims when he ordered them because they were a special...
-
Consider the function f and its graph. a. Estimate the zeros of the area function b. Estimate the points (if any) at which A has a local maximum or minimum. c. Sketch a graph of A, for 0 x 10,...
-
A normally distributed random variable has an unknown mean p, and a known variance 2 = 9. Find the sample size required to construct a 95 percent confidence interval on the mean that has total width...
-
The shelf life of a carbonated beverage is of interest. Ten bottles are randomly selected and tested, and the following results are obtained: Days 108 138 124 163 124 159 106 134 115 139 (a) We would...
-
Consider the shelf life data in Problem 2-5. Can shelf life be described or modeled adequately by a normal distribution? What effect would violation of this assumption have on the test procedure you...
-
How the best in details would theses table and graphs be described on Staff members' awareness of the Strategic Goals? Maximum level awareness (7) - Employee able to identify and articulate the...
-
What are some of the challenges a small medical office would have to transition from paper to an EHR system?
-
The use of pay grades and pay ranges and their relationship to internal alignment and external competitiveness. Why are pay grades and pay ranges necessary?

Study smarter with the SolutionInn App


