An adiabatic pistoncylinder system contains a 1000 W immersion heater and 4 kg of water. The water
Question:
An adiabatic piston–cylinder system contains a 1000 W immersion heater and 4 kg of water. The water is initially at 1 atm and 96% quality. The heater is operated for 7 minutes. Calculate the final volume of the system using Appendix A.3 for data.
Data From A.3
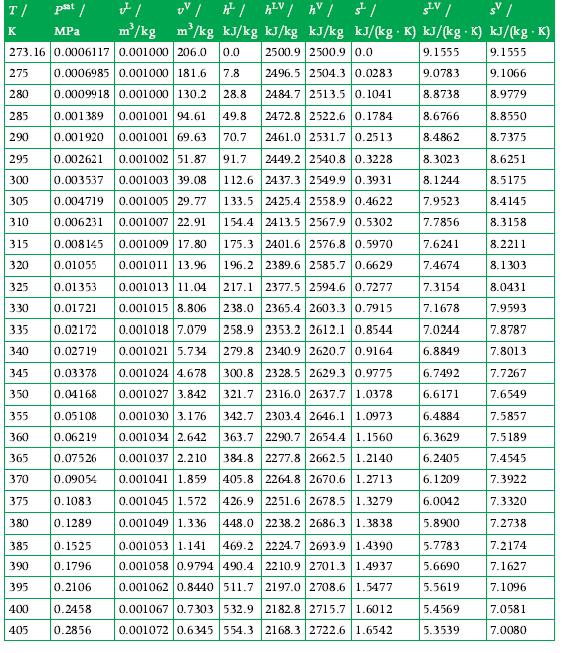
Transcribed Image Text:
T/ psat / K MPa UV /h²/h²V / hv / st/ SLV / s | m³/kg kJ/kg kJ/kg kJ/kg kJ/(kg. K) kJ/(kg K) kJ/(kg .K) 2500.9 2500.9 0.0 9.1555 9.1555 2496.5 2504.3 0.0283 9.0783 9.1066 2484.7 2513.5 0.1041 8.8738 8.9779 FI m²/kg 273.16 0.0006117 0.001000 206.0 0.0 275 0.0006985 0.001000 181.6 7.8 0.0009918 0.001000 130.2 28.8 280 285 0.001389 0.001001 94.61 49.8 2472.8 2522.6 0.1784 290 0.001920 0.001001 69.63 70.7 2461.0 2531.7 0.2513 295 0.002621 0.001002 51.87 91.7 2449.2 2540.8 0.3228 300 0.003537 0.001003 39.08 112.6 2437.3 2549.9 0.3931 133.5 2425.4 2558.9 0.4622 305 0.001005 29.77 0.004719 0.006231 0.001007 22.91 154.4 2413.5 2567.9 0.5302 310 175.3 2401.6 2576.8 0.5970 196.2 2389.6 2585.7 0.6629 217.1 2377.5 2594.6 0.7277 238.0 2365.4 2603.3 0.7915 258.9 2353.2 2612.1 0.8544 279.8 2340.9 2620.7 0.9164 315 0.008145 0.001009 17.80 320 0.01055 0.001011 13.96 0.01353 0.001013 11.04 0.01721 0.001015 8.806 335 0.02172 325 330 0.001018 7.079 0.02719 0.001021 5.734 340 345 0.03378 350 0.04168 355 0.05108 360 0.06219 365 0.07526 370 0.09054 375 0.1083 380 0.1289 385 0.1525 390 0.1796 395 0.2106 0.2458 400 405 0.2856 0.001024 4.678 300.8 2328.5 2629.3 0.9775 321.7 2316.0 2637.7 1.0378 0.001027 3.842 0.001030 3.176 2303.4 2646.1 1.0973 342.7 363.7 2290.7 2654.4 1.1560 0.001034 2.642 0.001037 2.210 0.001041 1.859 384.8 2277.8 2662.5 1.2140 405.8 2264.8 2670.6 1.2713 426.9 0.001045 1.572 2251.6 2678.5 1.3279 448.0 2238.2 2686.3 1.3838 0.001049 1.336 0.001053 1.141 469.2 2224.7 2693.9 1.4390 0.001058 0.9794 490.4 2210.9 2701.3 1.4937 0.001062 0.8440 511.7 2197.0 2708.6 1.5477 2182.8 2715.7 1.6012 0.001067 0.7303 532.9 0.001072 0.6345 554.3 2168.3 2722.6 1.6542 8.6766 8.4862 8.3023 8.1244 7.9523 7.7856 7.6241 7.4674 7.3154 7.1678 7.0244 6.8849 6.7492 6.6171 6.4884 6.3629 6.2405 6.1209 6.0042 5.8900 5.7783 5.6690 5.5619 5.4569 5.3539 8.8550 8.7375 8.6251 8.5175 8.4145 8.3158 8.2211 8.1303 8.0431 7.9593 7.8787 7.8013 7.7267 7.6549 7.5857 7.5189 7.4545 7.3922 7.3320 7.2738 7.2174 7.1627 7.1096 7.0581 7.0080
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
1 Make an idealized sketch of the system and identify the boundary indicating if it moves in time 2 ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
A vertical cylinder is fitted with a frictionless piston that is initially resting on stops. The cylinder contains carbon dioxide gas at 200 kPa, 300 K, and at this point the volume is 50 L. A...
-
A mixture of 4 kg of water vapor and 96 kg of dry air exists at 100 kPa and 25 8C in a 10-m3 volume. Determine the molar mass of the mixture and the volume occupied by the vapor. This represents very...
-
A 4.00-L sample of a diatomic ideal gas with specific heat ratio 1.40, confined to a cylinder, is carried through a closed cycle. The gas is initially at 1.00 atm and at 300 K. First, its pressure is...
-
Laverty Clinic plans to purchase a new centrifuge machine for its New York facility. The machine costs $94,000 and is expected to have a useful life of 6 years, with a terminal disposal value of...
-
The risk-free rate is currently 8.1%. Use the data in the accompanying table for the Fio familys portfolio and the market portfolio during the year just ended to answer the questions that follow. a....
-
In Figure three identical conducting spheres form an equilateral triangle of side length d = 20.0 cm. The sphere radii are much smaller than d, and the sphere charges are qA = ?? 2.00nC, qB = ??...
-
Identify the key elements of effective project cost management.
-
Citigroup Inc. (Citi) is a leading global financial services company with over 200 million customer accounts and operations in more than 140 countries. Its operating units Citicorp and Citi Holdings...
-
1. According to the theory of Gaussian distributions, what percentage of your measurements should be within 1 of the mean (see Figure 1)? ("Within 1 o" means the fraction of points within plus or...
-
Potassium, an alkali metal, enters a condenser in the vapor phase with a mass flow rate of 0.02 kg/s and at a temperature and pressure of 1370 K and 0.4 MPa. It exits as a saturated liquid at 0.2...
-
Your company asks you, the process engineer, to estimate the rate at which energy is transferred as heat through a steam turbine casing. This turbine operates with an inlet pressure of 16 bar and an...
-
Today is January 1. Forward prices for gold forward maturing on April 1 is $1,500 per ounce. The simple interest rate is 6 percent per year. What would be the forward price for a forward contract on...
-
Singh Company sold 75 units @ $350 each on October 31, Year 2. The following information is also available: Beginning inventory 25 units @ $175 February 2 purchase 20 units @ $180 June 15 purchase 45...
-
During April, Bloom Garden Supply Company experienced the following business events. The company uses the perpetual inventory system. 1) April 4 Purchased $10,000 of merchandise on account, terms...
-
How do environmental factors, including climate change, resource scarcity, and ecological degradation, shape patterns of social inequality, collective action, and global governance structures within...
-
The following is a summary of information presented on the financial statements of a company on December 31, 2025. Account Net Sales Revenue 2025 $609,000 2024 $503,000 Cost of Goods Sold 456,000...
-
How do sociological perspectives on race, ethnicity, and cultural identity intersect with processes of socialization, discrimination, and social mobility, particularly in the context of colonial...
-
The solubility of zinc oxalate, ZnC2O4, in 0.0150 M ammonia is 3.6 10-4 mol/L. What is the oxalate-ion concentration in the saturated solution? If the solubility product constant for zinc oxalate is...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
The oxidation of methane can take place according to either one of the following reactions: When the reactions stop at time tf because all the methane is burned, the total mass of the products (CO 2...
-
Acetylene (C 2 H 2 ) can be produced through a chemical reaction between water (H 2 O) and calcium carbide (C a C 2 ): where (s) and (l) indicate whether the substance is solid or liquid. A cave...
-
a) There are two isomers of butane: butane (C 4 H 10 ) and isobutane (methylpropane) (iso-C 4 H 10 ). Determine the standard enthalpy of isomerisation h of butane to isobutane in terms of the...
-
1. what should be debited for the Nov 15th transaction? 2. How much would the undadjusted trial balance be out by if the debit entry from the Nov. 2 entry was posted in error as $12,000? 3. What type...
-
Cullumber Company completes and transfers out 15,360 units and has 2,560 units of ending work in process that are 25% complete as to conversion costs. Materials are entered at the beginning of the...
-
Light Center, Final Project 6 $799,399.76 38,608.44 0,791.32 Accounting Services lab caps lock shift 4 5 Q W E R A S D F N 36 m control option command T Y 0 C V B Trans. 54 JJ The bank statement has...

Study smarter with the SolutionInn App


