Compute the standardized enthalpy and entropy of a mixture formed by an equal mass amount of CO
Question:
Compute the standardized enthalpy and entropy of a mixture formed by an equal mass amount of CO2?and H2O at 2 atm and 311 K. If liquid is present for these conditions, assume that the vapor phase can be approximated as an ideal gas mixture, and the liquid phase is pure water. Use:?
a) Values obtained from Table A.1, and?
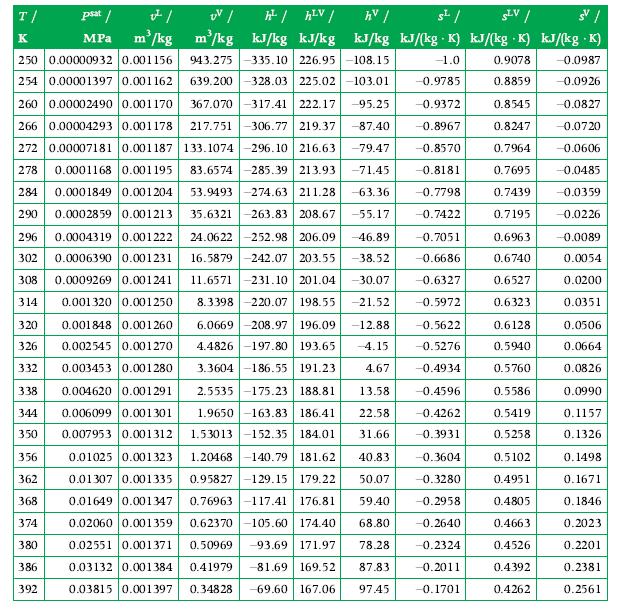
b) values obtained with GASMIX. If there is a difference, explain it.
Transcribed Image Text:
T/ psat / v²/ UV / h²/ hLV/ hv/ s²/ SLV/ SV/ K MPa m³/kg . m³/kg kJ/kg kJ/kg kJ/kg kJ/(kg K) kJ/(kg K) kJ/(kg .K) 943.275 -335.10 226.95-108.15 -1.0 250 0.00000932 0.001156 254 0.00001397 0.001162 260 0.00002490 0.001170 367.070-317.41 222.17 -95.25 639.200-328.03 225.02-103.01 -0.9785 -0.9372 -87.40 -0.8967 -79.47 -0.8570 -71.45 -0.8181 -63.36 -0.7798 -0.7422 266 0.00004293 0.001178 217.751-306.77 219.37 272 0.00007181 0.001187 133.1074 296.10 216.63 278 0.0001168 0.001195 83.6574 -285.39 213.93 284 0.0001849 0.001204 53.9493 -274.63 211.28 290 0.0002859 0.001213 35.6321-263.83 208.67 -55.17 296 0.0004319 0.001222 24.0622 -252.98 206.09 -46.89 302 0.0006390 0.001231 16.5879 -242.07 203.55 -38.52 308 0.0009269 0.001241 11.6571-231.10 201.04 -30.07 0.001320 0.001250 8.3398 220.07 198.55 -21.52 0.001848 0.001260 6.0669 -208.97 196.09 -12.88 326 0.002545 0.001270 4.4826 -197.80 193.65 -4.15 332 0.003453 0.001280 3.3604-186.55 191.23 338 0.004620 0.001291 314 320 4.67 2.5535-175.23 188.81 13.58 -0.4596 -0.4262 1.9650 -163.83 186.41 22.58 1.53013-152.35 184.01 31.66 -0.3931 344 0.006099 0.001301 350 0.007953 0.001312 356 0.01025 0.001323 0.01307 0.001335 0.01649 0.001347 -0.3604 362 -0.3280 1.20468-140.79 181.62 40.83 0.95827-129.15 179.22 50.07 0.76963-117.41 176.81 59.40 0.62370-105.60 174.40 68.80 368 -0.2958 374 0.02060 0.001359 -0.2640 380 0.50969 -93.69 171.97 78.28 -0.2324 0.02551 0.001371 0.03132 0.001384 0.41979 -81.69 169.52 87.83 386 -0.2011 392 0.03815 0.001397 0.34828 -69.60 167.06 97.45 -0.1701 -0.7051 -0.6686 -0.6327 -0.5972 -0.5622 -0.5276 -0.4934 0.9078 -0.0987 0.8859 -0.0926 0.8545 -0.0827 0.8247 -0.0720 0.7964 -0.0606 0.7695 -0.0485 0.7439 -0.0359 0.7195 -0.0226 0.6963 0.6740 0.6527 0.6323 0.6128 0.5940 0.5760 0.5586 0.5419 0.5258 0.5102 0.4951 0.4805 0.4663 0.4526 0.4392 0.4262 -0.0089 0.0054 0.0200 0.0351 0.0506 0.0664 0.0826 0.0990 0.1157 0.1326 0.1498 0.1671 0.1846 0.2023 0.2201 0.2381 0.2561
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a b From Appendix A1 we have MHo 001802 kgkmol and Mco 004401 kgkmol ...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
A thin (1.0-mm-thick) coat of fresh paint has just been sprayed over a 1.5-m by 1.5-m square steel body part, which approximates a flat surface. The paint contains a volatile solvent that initially...
-
What is meant by Cumulative Record Card?
-
Briefly explain the Nature and purpose of the Cumulative Record Card (CRC) along with the design and advantages of the CRC.
-
Find the radius of convergence and interval of convergence of the series. 00 (-1)"x" n=0 n + 1
-
Nordau and Boyer (2000) estimated that the United States would bear over 90% of the total world cost of achieving the Kyoto targets for greenhouse gas emission reductions. Explain how this can be,...
-
Lukow Products is investigating the purchase of a piece of automated equipment that will save $400,000 each year in direct labor and inventory carrying costs. This equipment costs $2,500,000 and is...
-
During the normal operating period, the deterioration of a machine usually a. decreases b. increases c. remains constant
-
Relaxing Recliner Chairs completed the following selected transactions: 2011 Jul 1 Sold inventory to Great Mart, receiving a $45,000, nine-month, 12% note. Ignore cost of goods sold. Oct 31 Recorded...
-
What about global supply chain uncertainty, risk and probabilities, hazards and gain insight into a variety of industries' risk. . A case study analysis requires you to investigate a business...
-
Which series has the highest beta. BraveNewCoin Liquid Index for Bitcoin 1D BNC Trading Brave Ne Yellow Green Blue Orange
-
Obtain the expression for the specific flow exergy (9.5) from its definition.
-
Derive the reference I-law cycle efficiency for the evaluation of the II-law efficiency of refrigeration systems.
-
A public water supply was found to contain 0.8 part per billion (ppb) by mass of chloroform, CHCl 3 . (a) How many CHCl 3 molecules would be present in a 350 mL glass of this water? (b) If the CHCl 3...
-
After many years of international consulting and trading activities with Hong Kong and China, Klaus Koehler established a services company, the Klako Group, in Hong Kong in 1979. The company provided...
-
Three things set The Pampered Chef, a direct selling company, apart from its competition in the housewares industry: a passion for providing unique, quality products, a dedication to ensuring...
-
Select an industrially developed country (perhaps Japan or a European country) and a relatively less developed country (perhaps a Latin American or African country). Contrast the relative importance...
-
In 1943 a 17-year-old Swedish boy started what was to become a multibillion euro company by selling work pants and other farm supplies door-to-door. Ingvar Kamprad began selling farm implements under...
-
Mrs Kubo is the mother of an autistic child who fortunately was diagnosed when still very young. In order to help him develop word skills when he was still a small baby, Mrs Kubo started cutting out...
-
All of the following processes involve a separation of either a mixture into substances or a compound into elements. For each, decide whether a physical process or a chemical reaction is required. a....
-
Open Text Corporation provides a suite of business information software products. Exhibit 10-9 contains Note 10 from the companys 2013 annual report detailing long-term debt. Required: a. Open Text...
-
Explain why the solubility of an ionic compound increases as the ionic strength of the solution increases (at least up to ~ 0.5 M).
-
Which statements are true? In the ionic strength range 00.1 M, activity coefficients decrease with (a) Increasing ionic strength; (b) Increasing ionic charge; (c) Decreasing hydrated radius.
-
Calculate the activity coefficient of Al 3+ when = 0.083 M by linear interpolation in Table 7-1. Table 7-1 Ion size Ionic strength (p. M) Ion (a, pm) 0.001 0.005 0.01 0.05 0.1 Charge = +1 H*...
-
Agnessa is 23 years old and receives $350 per month from a trust fund. If she decides to invest this money at the end of every 3 months into an investment paying 6.75% compounded quarterly, how much...
-
Long issued 5,000 shares of stock for $10,000. Long used $2,000 of the proceeds to purchase supplies, and borrowed $8,000 cash from the bank. What are Long's total assets ?
-
A project requires an initial, up-front (at t=0) capital expenditure of $11,820. It then generates constant annual cash inflows for the next 21 years of $800 with the first payment due at t=1. After...

Study smarter with the SolutionInn App


