Oxygen flows through an adiabatic steady flow compressor as saturated vapor at a rate of 1000 kg/hr.
Question:
Oxygen flows through an adiabatic steady flow compressor as saturated vapor at a rate of 1000 kg/hr. The saturated vapor enters at 2.5 bar and exits at 17.5 bar and 175 K. Calculate the shaft work per unit mass of oxygen and the required power to drive the compressor. Use Appendix A.8 for data.
Data From A.8
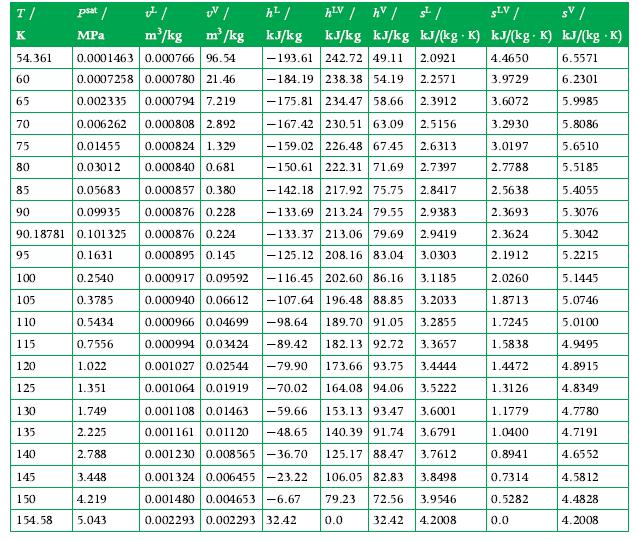
Transcribed Image Text:
JI m³/kg m³/kg 54.361 0.0001463 0.000766 96.54 0.0007258 0.000780 21.46 0.002335 0.000794 7.219 0.006262 0.01455 0.03012 0.05683 0.09935 0.101325 0.1631 T/ K 60 65 70 75 80 85 90 90.18781 95 100 105 110 115 120 125 130 135 140 psat / MPa 1.749 2.225 2.788 3.448 4.219 154.58 5.043 145 150 0.2540 0.3785 0.5434 0.7556 1.022 1.351 h²/ kJ/kg -193.61 242.72 49.11 2.0921 -184.19 238.38 54.19 2.2571 -175.81 234.47 58.66 2.3912 -167.42 230.51 63.09 2.5156 -159.02 226.48 67.45 2.6313 -150.61 222.31 71.69 2.7397 -142.18 217.92 75.75 2.8417 -133.69 213.24 79.55 2.9383 -133.37 213.06 79.69 2.9419 -125.12 208.16 83.04 3.0303 -116.45 202.60 86.16 3.1185 196.48 88.85 3.2033 189.70 91.05 3.2855 182.13 92.72 3.3657 173.66 93.75 3.4444 164.08 94.06 3.5222 153.13 93.47 3.6001 140.39 91.74 3.6791 3.7612 125.17 88.47 106.05 82.83 3.8498 79.23 0.0 72.56 3.9546 32.42 4.2008 hLV / hv / s² / SLV/ 1 kJ/kg kJ/kg kJ/(kg K) kJ/(kg. K) kJ/(kg .K) 4.4650 6.5571 3.9729 6.2301 3.6072 5.9985 0.000808 2.892 0.000824 1.329 0.000840 0.681 0.000857 0.380 0.000876 0.228 0.000876 0.224 0.000895 0.145 0.000917 0.09592 0.000940 0.06612 -107.64 0.000966 0.04699 -98.64 0.000994 0.03424 -89.42 0.001027 0.02544 -79.90 0.001064 0.01919 -70.02 0.001108 0.01463 -59.66 0.001161 0.01120 -48.65 0.001230 0.008565-36.70 0.001324 0.006455 -23.22 0.001480 0.004653-6.67 0.002293 0.002293 32.42 3.2930 3.0197 2.7788 2.5638 2.3693 2.3624 2.1912 2.0260 1.8713 1.7245 1.5838 1.4472 1.3126 1.1779 1.0400 0.8941 0.7314 0.5282 0.0 5.8086 5.6510 5.5185 5.4055 5.3076 5.3042 5.2215 5.1445 5.0746 5.0100 4.9495 4.8915 4.8349 4.7780 4.7191 4.6552 4.5812 4.4828 4.2008
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Mh comp Mh Assuming the that the kinetic energy of the fluid at the inlet and outlet of the compress...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
a) A steam turbine operating at a steady state has water entering at 3 MPa, 500 C, and a velocity of 10 m/s. The water exits at 800 kPa, 250 C, and 100 m/s. Find the (a) specific work (kJ/kg) and the...
-
An air-conditioner with refrigerant-134a as the working fluid is used to keep a room at 23°C by rejecting the waste heat to the outdoor air at 34°C. The room gains heat through the walls and...
-
An air-conditioner with refrigerant-134a as the working fluid is used to keep a room at 26°C by rejecting the waste heat to the outdoor air at 34°C. The room gains heat through the walls and...
-
The Howell Corporation has the following account balances (in millions): Required Prepare an income statement and a supporting schedule of cost of goods manufactured for the year ended December 31,...
-
Using the data in the following table, assume you are using a constant-dollar plan with a rebalancing trigger of $1,500. The stock price represents your speculative portfolio, and the MM mutual fund...
-
In Figure the particles have charges q1 = ?? q2 = 100nC and q3 = ?? q4 = 200nC, and distance a = 5.0cm. What are the(a) x and(b) y components of the net electrostatic force onparticle? t (2 $ 2) 1
-
Define cost estimation and discuss its importance in project planning.
-
Atlantic Airlines is considering these two alternatives for financing the purchase of a fleet of airplanes: 1. Issue 50,000 shares of common stock at $40 per share. (Cash dividends have not been paid...
-
From the intensity function for two point sources, the location and intensity of the peak can easily be calculated. It is very different for the intensity function of a single finite-width slit in...
-
In a coal-fired power station, steam (H 2 O) enters the nozzle in a turbine at T 1 = 800 K, P 1 = 2 MPa at negligible velocity, and emerges at high velocity as a liquidvapor mixture having quality x...
-
Potassium, an alkali metal, enters a condenser in the vapor phase with a mass flow rate of 0.02 kg/s and at a temperature and pressure of 1370 K and 0.4 MPa. It exits as a saturated liquid at 0.2...
-
Svahn, AB, is a Swedish manufacturer of sailing yachts. The company has assembled the information shown below that pertains to two independent decision-making contexts called Case A and Case B: Case...
-
During the year, in Sigma Corporation, Job P90 was started, completed, and sold to the customer for $4.000 The plantwide predetermined overhead rate was $20 per machine hours. The following...
-
What is the difference between autotrophs and heterotrophs? Please give examples.
-
A common symptom of pharyngitis is swelling of the anterior cervical lymph nodes. Why may lymph nodes swell in the presence of an infection? what else may cause swollen lymph nodes? (hint: What part...
-
Marie is a single taxpayer that operates a small shop in Boston. The business operates as a sole proprietorship with no employees. Marie's Schedule C reports income of $90,000. Her taxable income is...
-
A $100,000 investment today has a $400,000 value in 3 years. What is the annual rate of return for this investment?
-
Ammonia, NH3, is a base that ionizes to give NH4+ and OH (Kb = 1.8 10-5). You add magnesium sulfate to an ammonia solution. Calculate the concentration of Mg2+ ion when magnesium hydroxide, Mg(OH)2,...
-
What is a make-or-buy decision?
-
At sea level, water from the outlet of a river is diverted to a power plant that operates on the principle of osmosis. A turbine is installed in the pipe that brings the river water to an osmotic...
-
A container at a pressure p and temperature T contains two substances 1 and 2, present both in liquid and gas phases. Estimate the partial pressure p A of substance Entropy of mixing, as a function...
-
The variance v is the number of degrees of freedom of a system consisting of r substances in m phases taking part in n chemical reactions. The variance v is obtained by subtracting n constraints from...
-
What is the step in the speechmaking process when the speaker studies the potential listeners through techniques such as interviews and questionnaires?
-
What should Lisa do ? Should she still keep the money and look the other way? Does the source of the money matter or does the end justify the means?
-
State in what circumstances you have assisted in , or demonstrated awareness of the selection of save and sustainable materials, components and systems and have identified risk and applied risk...

Study smarter with the SolutionInn App


