Suggest a structure for the compound whose mass spectrum is asfollows: 100 107 81 93 188 70
Question:
Suggest a structure for the compound whose mass spectrum is asfollows:
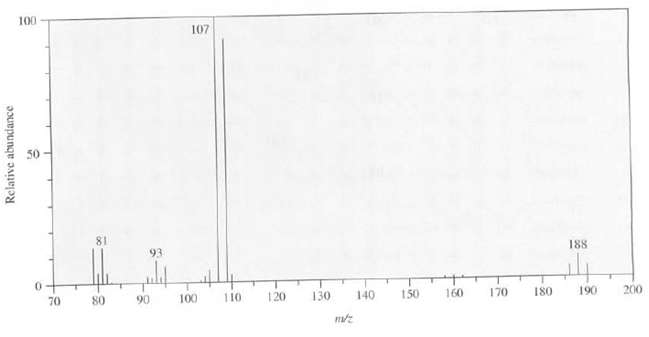
Transcribed Image Text:
100 107 81 93 188 70 80 110 120 90 100 130 150 160 170 140 180 190 200 Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The peaks at mz 186 188 and 190 in a 121 ratio suggests the pres...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Suggest a composition for the halogen compound whose mass spectrum is shown in the figure. Assign each of the major peaks. 100 127 173 160 25 158 162 7 48 HT 79 91 160 170 208 35 160 173 IITI'T1 30...
-
Suggest a structure for the compound (4). Using curly arrow notation, draw out the mechanism for the formation of compound (4). Use 3-dimensional structures to rationalize the relative...
-
Suggest a structure for the compound (5). Using curly arrow notation, draw out the mechanism for the conversion of compound (5) into compound (6). Use 3-dimensional structures to rationalize the...
-
What is control resolution in a robot positioning system?
-
1. Explain the pros and cons of the politics- administration dichotomy as espoused by Woodrow Wilson 2. Discuss the various uses of public budget
-
How would you suggest that your department apply these priorities in planning its management training programs?
-
For each of the following sets of data, (1) calculate the mean of the scores \(\left(\mathrm{X}^{-} ight),(2)\) calculate the deviation of each score from the mean \(\mathrm{X}-\mathrm{X}^{-}\), and...
-
The following selected transactions are from Springer Company. 2012 Nov. 1 Accepted a $ 4,800, 90-day, 8% note dated this day in granting Steve Julian a time extension on his past-due account...
-
Felix & Company reports the following information. Period Units Produced Total Costs 1234567890 0 640 1,040 $ 4,740 4,040 4,440 1,440 5,040 1,840 4,140 2,240 4,340 2,640 8,840 3,040 16,240 3,440...
-
Using the Gibbs function data, determine the equilibrium constant KP for the reaction H2 + 1/2 O2 H2O at (a) 298 K and (b) 2000 K. Compare your results with the KP values listed in Table A28.
-
Explain how the peaks at m/z 115, 101, and 73 arise in the mass spectrum of 3-methy-3-heptanol.
-
The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound, what is the structure of the ion responsible for the peak at m/z 43...
-
Solve the preceding problem for the following data: (x = 420 ( 10-6, (y = - 170 ( 10-6, Yxy = 310 ( 10-6, and ( = 37.5o.
-
mr spahn fills up his gas tank in iceland where the currency used is iclandic krona ( isk ) . One us dollar is equivalent to 1 4 4 krona ( $ 1 = 1 4 4 Krona ) and gasoline costs 3 2 5 krona per liter...
-
Find the attractive force ( in newtons ) between a 6 . 7 0 x 1 0 - 5 coulomb point charge and a - 7 . 7 9 x 1 0 - 5 coulomb point charge that are separated by 0 . 0 7 meters.
-
A rocket engineer pumps liquid propane and hydrogen to a rocket engine. The rocket engine is filled up completely. Then, rocket is ignited and accelerated up to the sky in a very fast way. Please...
-
Two masses have the same charge, 3 0 microColoumbs. One is placed at ( 0 , 3 m ) and the other is placed at ( 4 m , 0 ) , so they are on the corners of a 3 - 4 - 5 triangle. a . Find the magnitude of...
-
A spaceship orbits Planet X at a distance of 1.5x107m from the planet's center. a) If the orbital period is 6 hours (21,600s), what is the mass of Planet X? b) If the spaceship wants to leave Planet...
-
Quantitative Risk Assessment. Assume the following information for an IT project: Calculate the overall risk factor for this project. Would you assess this level of risk as low, moderate, or high?...
-
A researcher reports a significant two-way between-subjects ANOVA, F(3, 40) = 2.96. State the decision to retain or reject the null hypothesis for this test.
-
What did investors in Treasury bonds expect the Federal Reserve to do with interest rates?
-
When two substituents are on the same side of a ring skeleton, they are said to be cis, and when on opposite sides, trans (analogous to use of those terms with 1,2-disubstituted alkene isomers)....
-
Examine the diagram showing an a-helical protein structure in Section 2.13E. Between what specific atoms and of what functional groups are the hydrogen bonds formed that give the molecule its helical...
-
Using a three-dimensional formula, show the direction of the dipole moment of CH3OH. Write (+ and (- signs next to the appropriate atoms.
-
S&J Catering is a small catering business operating in Western Sydney. Business partners, Jack and Simon, established the business a year ago. To start up the business, the partners contributed...
-
In 20X1, Lee was hurt in the course of his employment with Foster Farms. In 20X1, Lee was paid the following amounts from Foster Farms' worker's compensation insurance company: $30,000 for a physical...
-
Briefly describe tax administrative matters including: Tax collection and withholding mechanisms (PAYG withholding, PAYG instalments).

Study smarter with the SolutionInn App


