Suggest syntheses of these amino acids from the indicated starting materials: NH, Br a) PHCH,CHCO,H from PHCH,CHCO,H
Question:
Suggest syntheses of these amino acids from the indicated starting materials:
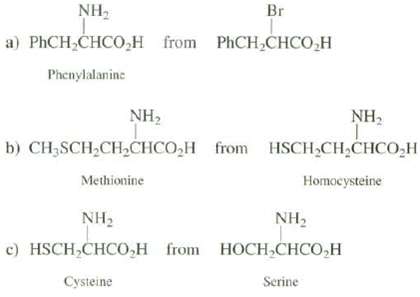
Transcribed Image Text:
NH, Br a) PHCH,CHCO,H from PHCH,CHCO,H Phenylalanine NH2 NH, b) CH3SCH2CHCHCO,H from HSCH CH,CHCO,H Methionine Homocysteine NH2 с) HSCH,CHCO,Н гom NH2 HОСH,CНCO,H Cysteine Serine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
Br a PhCHCHCOH excess NH3 NH ...View the full answer

Answered By

Marcus Solomon
I am committed to ensuring that my services always meet the clients' expectations.
4.60+
82+ Reviews
117+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary. Your syntheses may produce both enantiomers of any target that ischiral. , CH,CH,CH3 a)...
-
Propose syntheses of the following molecules from the indicated starting materials. Make use of any other reagents or solvents that you need. In some cases, there may be no alternative but to employ...
-
Show how Tagamet could be synthesized from the indicated starting materials. CH2OH CH CH SCH2CH2NH NHCH N CH CHS SCH CH3 +CH:NH2
-
Suggest two strategies for the company to minimise the impact of the strike on business operation?
-
In your own words, explain the difference between cost of goods manufactured and cost of goods sold.
-
When a group of people must decide whether to buy a shared public good or service, the free-rider problem frequently occurs because: a. People have an incentive to understate how much the facility is...
-
When creating association rules, why is it common to only consider rules with a confidence measure above a minimum threshold? a. A minimum confidence threshold is not necessary if item granularity is...
-
Refer to Cornerstone Exercise 7.3. Now assume that Valron Company uses the sequential method to allocate support department costs. The support departments are ranked in order of highest cost to...
-
K A country's savings S is defined implicitly in terms of its national income I by the equation where both S and I are in billions of dollars. Find the marginal propensity to consume when 1-22 and S...
-
QED Electronics Company had the following transactions during April while conducting its television and stereo repair business. 1. A new repair truck was purchased for $ 19,000. 2. Parts with a cost...
-
Another Diphenhydramine synthesis is shown in the following equation: (a) Show a mechanism for the first step in this synthesis. (b) Explain which mechanism is occurring in the secondstep. OCH CH...
-
(a) Explain which of the following two products is formed when the reactant alkyl chloride reacts with sodium acetate in DMSO. (b) Explain which of the following two product alcohols is formed when...
-
The following table represents the market share (in number of movies, gross in millions of dollars, and millions of tickets sold) of each type of movie in 2016: a. Construct a bar chart, a pie chart,...
-
Suppose we find that multinational firms are paying much lower wages in some poor countries than they would have to pay in the United States. Would this be sufficient evidence that these firms are...
-
What is the WTO? Where is it located, and what does it do?
-
What is the difference between fair and free trade?
-
There were several major international financial crises in the 1990s. What role did globalization play in these crises?
-
The chi square distribution with 4 degrees of freedom is given by \[f(x)= \begin{cases}\frac{1}{4} \cdot x \cdot e^{-x / 2} & x>0 \\ 0 & x \leq 0\end{cases}\] Find the probability that the variance...
-
In this exercise, we explore the relationship between total household expenditures and expenditures on clothing. Use the data file malawi_small (malawi has more observations) and observations for...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
Solve each equation. 8 2 =x + 3 5 + 23 11 -x+ 5 45 6x + 7 3
-
Pentalene is a most elusive molecule and has never been isolated. The pentalene dianion, however, is well known and quite stable. Explain. 12- Pentalene Pentalene dianion
-
Indole is an aromatic heterocycle that has a benzene ring fused to a pyrrole ring. Draw an orbital picture of indole. (a) How many ? electrons does indole have? (b) What is the electronic...
-
Ribavirin, an antiviral agent used against hepatitis C and viral pneumonia, contains a 1, 2, 4-triazole ring. Why is the ringaromatic? 1,2,4-Triazole ring N- NH2 N-N Ribavirin OH
-
www The sugar cane industry is a big industry on Hawaii. Raw sugar cane is first cut from the fields, then chopped and shredded. The raw cane contains 15 wt% sucrose, 25 wt% solids, and water, along...
-
What does a manager who subscribes to Theory X believes ? Explain thoroughly.
-
Keri, in forming a new corporation, transfers land to the corporation in exchange for 100 percent of the stock of the corporation. Keri's basis in the land is $295,000, and the corporation assumes a...

Study smarter with the SolutionInn App


