Sulfuric acid is the chemical produced in the United States with the highest volume of production. In
Question:
Sulfuric acid is the chemical produced in the United States with the highest volume of production. In one of the earliest processes used to make it, an ore containing iron pyrites (FeS2) is roasted (burned) with air. The following reactions take place in the roasting furnace: The gas leaving the reactor goes to a catalytic converter in which most of the SO2 produced is further oxidized to SO3: Finally, the gas leaving the converter passes through an absorption tower, in which the SO3 is absorbed in water to produce sulfuric acid (H2SO4).
(a) An ore containing 82 wt% FeS2 and 18% inerts is fed to a roasting furnace. Dry air is fed to the furnace in 40% excess of the amount theoretically required to oxidize all of the sulfur in the ore to SO3, an FeS2 oxidation of 85% is obtained, with 40% of the FeS2 converted forming sulfur dioxide and the rest forming sulfur trioxide. Two streams leave the roaster: a gas stream containing SO2, SO3, 02, and N2, and a solid stream containing unconverted pyrites, ferric oxide, and inert material in the ore. Calculate the required feed rate of air in standard cubic meters per 100 kg of ore roasted and the molar composition and volume (SCMJ100 kg of ore) of the gas leaving the roasting oven.
(b) The gas leaving the roasting oven enters the catalytic converter, which operates at 1 atm. The conversion reaction [3] proceeds to an equilibrium point at which the component partial the gases are first heated to 600?C, at which Kp = 9.53 atm?1/2, and are then cooled to 400?C, where K,, = 397 atm?1/2. The rate of the forward reaction increases sharply with temperature and is several orders of magnitude greater at 600?C than at 400?C. Calculate the equilibrium fractional conversions of sulfur dioxide in the converter when the temperature is 600?C and when it is 400?C. Briefly explain why the converter gases are initially heated and then cooled.
(c) Assuming complete conversion to sulfuric acid of the sulfur trioxide leaving the converter, how many kg of H2SO4 will be produced per kg of sulfur in the ore? What would this ratio have been if all of the sulfur in the ore had been converted? Summarize the factors responsible for the second number being larger than the first one.
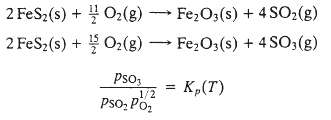
Step by Step Answer:

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau





