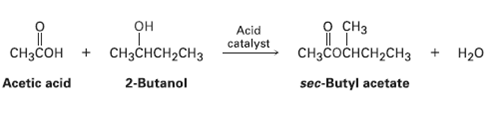
Suppose that acetic acid (CH3CO2H) reacts with (S)-2-butanol to form an ester (see Worked Example 9.6). What
Question:
Suppose that acetic acid (CH3CO2H) reacts with (S)-2-butanol to form an ester (see Worked Example 9.6). What stereochemistry would you expect the product(s) to have? What is the relationship of theproducts?
Transcribed Image Text:
о сHз CHзCоснсH2сHз Acid он CH3CHCH2CH3 catalyst CHзcон Нао 2-Butanol Acetic acid sec-Butyl acetate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
HC COH HOC is Acetic acid CH3 acid catalyst CHCH3 S...View the full answer

Answered By

Rinki Devi
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and helped them achieve great subject knowledge.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Would you expect the 2-octanol formed by SN2 hydrolysis of (+) - 2-bromooctane to be optically active? If so, what will be its absolute configuration and sign of rotation? What about the 2-octanol...
-
Would you expect the substituent N (CH3)3 to more closely resemble N (CH3)2 or NO2 in its effect on rate and regioselectivity in electrophilic aromatic substitution? Why?
-
Would you expect the temperature of a liquid to change as it is throttled? Explain.
-
Sarah is confused about the difference between the perimeter and the area of a polygon. Explain the two concepts and the distinction between them.
-
Air-conditioning units contain no water whatever, yet it is common to see water dripping from them when they're operating on a hot day? Discuss.
-
Recall that TCP can be enhanced with SSL to provide process-to-process security services, including encryption. Does SSL operate at the transport layer or the application layer? If the application...
-
Sports Consulting Pty Ltd is a company set up by sports and recreation management students to gain experience in running their own business. It had the following data for the year ended 2025....
-
For each of the following situations, determine whether the company should (a) Report a liability on the balance sheet, (b) Disclose a contingent liability, or (c) Not report the situation. Justify...
-
Your grandfather has an annuity that is worth $ 9 9 3 8 4 today. It will give him monthly payments for 2 4 years. If the rate is 4 . 4 5 % , what will be his monthly payment? Show work and formula
-
A $1,000 par bond with an annual coupon has only one year until maturity. Its current yield is 6.713%, and its yield to maturity is 10%. What is the price of the bond?
-
Does the following structure represent a meso compound? If so, indicate the symmetryplane.
-
What stereoisomers would result from reaction of ()-lactic acid with (S)-1-phenyl- ethylamine, and what is the relationship between them?
-
Show that roasting lead (2PbS + 3O 2 ? 2 PbO +2 SO 2 ) is an exothermic reaction and compute the free energy of this reaction under standard conditions. See Table for data. Compound AG (kJ/mol)...
-
Find the first order partial derivative f(x,y) of the function f(x,y)=9x3+ - 4xy - 3
-
The current market price of a $ 1000 par bond is $ 1 , 025. Its semiannual coupon rate is 7 % and it matures in 5 years. What is the pretax cost of debt? What is the after-tax cost of debt using the...
-
a) b) 308 Suppose SSH Inc. is currently $12 per share and will go either up to $16 or down to $10. Suppose the risk-free interest rate is 3%. We are interested in finding the price of a put option....
-
Use Newton's method to approximate a root of the equation 3x7 + 4x4 + 2 = 0 as follows. Let x1 = 2 be the initial approximation. The second approximation 2 is and the third approximation 23 is Carry...
-
You have done the Innovation Report and recommended supporting an inventive workplace, and You emailed the report to your manager and the company's owners for review. A meeting has been planned with...
-
What would be an example of how Televerde might implement a high-involvement work practice to complement what it currently does?
-
Is times interest earned meaningful for utilities? Why or why not?
-
A sample of gas has a mass of 0.311 g. Its volume is 0.225 L at a temperature of 55 C and a pressure of 886 mmHg. Find its molar mass. SORT The problem gives you the temperature and pressure of...
-
Give the product expected when methyl methacrylate (methyl 2-methylpropenoate) reacts with each of the following reagents. (a) C2H5SH and NaOMe catalyst in MeOH (b) NaOH
-
Give a curved-arrow mechanism for each of the following reactions. H2C NaOEt catalyst O Ph SH EtOH CH PhSH2C O (mixture of stereoisomers; why?) CH3
-
Provide structures for the missing nucleophiles that could be used in the following transformations. NaOEt Ho+ CH, CH3
-
What are the vulnerabilities of embedded operating systems, such as, but not limited to, IoT devices, programmable logic devices, and vehicle control systems.
-
Factor the polynomial completely. 7x-17x-12
-
What are some relevant recommendations about Coca-Cola's Planning, Design, and Implementation of an Enterprise Resource Planning System that other companies can apply to their own initiatives?

Study smarter with the SolutionInn App


