Tamoxifen is a drug used in the treatment of breast cancer, how would you prepare tamoxifen from
Question:
Tamoxifen is a drug used in the treatment of breast cancer, how would you prepare tamoxifen from benzene, the following ketone, and any other reagents needed?
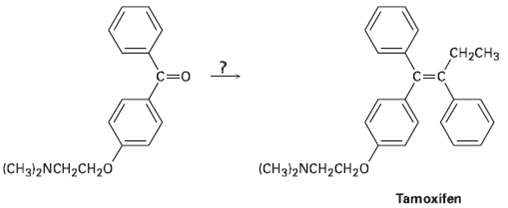
Transcribed Image Text:
CH2CH3 C=0 C=c (CH3!2NCH2CH20 (CH3)2NCH2CH20 Tamoxifen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 52% (17 reviews)
i CH3CHCCI AICI3 1 Br FeBr3 2 Mg ether OH CHCHCH3 PBr3 CCHCH3 MgBr Br 1 LIAIH4 ...View the full answer

Answered By

Moses mwangi
With prior writing experience, be sure that I will give a great grade, If not an A+, it will be something close to this. My reviews speaks it all, Try me!!
4.80+
78+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Tamoxifen is a drug often used to treat breast cancer patients. One effect of the drug is to change the levels of cortisol-binding globulin (CBG). One study attempted to see if the effect of...
-
How would you prepare o-hydroxyphenyl-acetaldehyde from phenol? More than one step is required. HO o-Hydroxyphenylacetaldehyde CH-CO
-
How would you prepare pentanal from the following starting materials? (a) CH 3 CH 2 CH 2 CH 2 CH 2 OH (b) CH 3 CH 2 CH 2 CH 2 CH = CH 2 (c) CH 3 CH 2 CH 2 CH 2 CO 2 CH 3
-
Use a linear interpolation to estimate properties of ammonia to fill out the table below P [kPa] T [ C] v [m3/kg] x a) 550 0.75 b) 80 20 c) 10 0.4
-
1. Do you think the model used by Seats2Meet would work in the United States? Why or why not? 2. In what ways can social capital help you in running a start-up firm or doing freelance projects?
-
The units of an item available for sale during the year were as follows: There are 48 units of the item in the physical inventory at December 31. The periodic inventory system is used. Determine the...
-
A study has three experimental units and two treatments-A and B. List all possible treatment assignments for the study. How many are there? In general, show that there are $2^{N}$ possible treatment...
-
Which of these two bonds offers the highest current yield? Which one has the highest yield to maturity? a. A 6.55 percent, 22-year bond quoted at 52.000 b. A 10.25 percent, 27-year bond quoted at...
-
A uniform beam of length 10 m and mass 100 kg is supported by pylons on either end. A 200 kg box is centered at 5 m from the left end of the beam. What is the downward force on the left most pylon?
-
Engineers are testing company fleet vehicle fuel economy (miles per gallon) performance by using different types of fuel. One vehicle of each size is tested. Does this sample provide sufficient...
-
When Cyclohexanone is heated in the presence of a large amount of acetone cyanohydrin and a small amount of base, Cyclohexanone cyanohydrin and acetone arc formed. Propose amechanism. HO CN OH "OH +...
-
Paraldehyde, a sedative and hypnotic agent, is prepared by treatment of acetaldehyde with an acidic catalyst. Propose a mechanism for the reaction. . C H+ H catalyst C Paraldehyde
-
Solve the given systems of equations using the inverse of the coefficient matrix. 6x + 6y 3z = 24 x + 4y + 2z = 5 3x 2y + z = 17
-
Explain to a new manager the Family and Medical Leave Act. Explain the law and then give him a few examples. Talk about what you can take FMLA for and how long you can be away. include some history...
-
What is the best way to establish a will that will contribute to family cohesiveness after both parents are gone? Why is family cohesiveness threatened when parents die?
-
Could you elaborate on the principles of aspect-oriented programming (AOP) and its applications in cross-cutting concerns management within software systems?
-
Reconciliation is an accounting procedure that compares two sets of records to check that the figures are correct and in agreement. Reconciliation also confirms that accounts in ageneral ledgerare...
-
Answers must reference common law decisions and/or sections in the relevant legislation Generally, under what circumstances, will there be an intention to create legal relations when work is...
-
Let \(\left\{X_{n}ight\}_{n=1}^{\infty}\) be a sequence of independent and identically distributed random variables following a \(\operatorname{ChiSquared}(\theta)\) distribution. A saddlepoint...
-
Compile data on consumption and expenditures for the following categories in 30 different countries: (1) food and beverages, (2) clothing and footwear, (3) housing and home operations, (4) household...
-
Use the VSEPR theory to predict the shapes of the anions (a) ClO 4 - ; (b) S 2 O 3 2- (that is, SSO 3 2- ); (c) PF 6 - ; (d) I 3 - .
-
The acid-catalyzed aldol condensation of acetone (just shown) also produces some 2, 6-dimethylhepta-2, 5-dien-4-one. Give a mechanism that explains the formation of this product.
-
Heating acetone with sulfuric acid leads to the formation of mesitylene (1, 3, 5-trimethylbenzene). Propose a mechanism for this reaction.
-
(a) Provide a mechanism for the aldol addition of propanal shown here. (b) How can you account for the fact that the product of the aldol addition is 3-hydroxy- 2-methylpentanal and not...
-
The Power of Trade and Comparative Advantage: Work It Out 3 ? Here's another specialization and exchange problem. This problem is wholly made-up, ? so that you won't be able to use your intuition...
-
Government spending as a fiscal policy tool is used to: A) ?Decrease the national debt B) ?Directly stimulate economic activity by increasing demand C) ?Reduce inflation D) ?Lower interest rates
-
Automobile demand has fallen during a recent recession, and Ana has been laid off from her job on the assembly line. Rajiv recently lost his job as a waiter at a local restaurant. A recent increase...

Study smarter with the SolutionInn App


