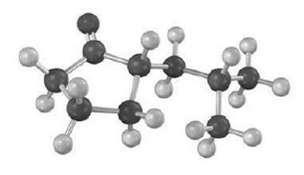
Tell the prominent IR absorptions and mass spectral peaks you would expect for the following compound:
Question:
Tell the prominent IR absorptions and mass spectral peaks you would expect for the following compound:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (20 reviews)
IR The only important IR absorption for the compound is seen at 1750 cm 1 wher...View the full answer

Answered By

Saurabh Tripathi
I'm first division graduate in Mechanical and Automation Engineering/Mechanical Engineering. I'm preparing for competitive examinations as GATE & UPSC Engineering Services Examinations and appeared this year before that I have been closely following various national level competitive examinations and know about various tips and tricks needed to excel it. I love solving problems related to Mechanical Engineering and apart from that I would also like to help students in High school/Intermediate Physics and Mathematics problems.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the NMR spectra you would expect for the following compounds. (a) (CH3)2CH-O-CH(CH3)2 (b) (c) Ph-CH(CH3)2 (d) (e) CI-CH2-CHC_O_CH3 - CH2COOCH CH CH2 COOCH2CH3
-
Describe the 1H NMR spectrum you would expect for each of the following compounds, using relative chemical shifts rather than absolute chemical shifts: a. BrCH2CH2Br b. CH3OCH2CH2CH2Br c. d. e. f. g....
-
List the different intermolecular forces you would expect for each of the following compounds: a. Propanol, CH3CH2CH2OH b. (See model below) c. Sulfur dioxide, SO2
-
Suppose that a countrys annual growth rates were 5, 3, 4, 1, 2, 2, 3, 4, 6, and 3 in yearly sequence over a 10-year period. What was the countrys trend rate of growth over this period? Which set of...
-
How do age, marital status, household size, employment situation, and other personal factors affect financial planning?
-
It is estimated that in 2030, there will be a total of 4.072 million knee replacement and hip replacement surgeries, with knee replacements outnumbering hip replacements by 2.928 million (Source:...
-
Describe the Agile Development Model, its characteristics, and shortcomings.
-
Home Entertainment is a small, family-owned business that purchases LCD televisions from a reputable manufacturer and sells them at the retail level. The televisions sell, on average, for $1,500...
-
Evaluate the critical components to reduce turnover and increase employee retention. very important, no short answers very important to give details very important to provide an explanation very...
-
Part 1: 1. If Wilmington wishes to sue Santiago, what court or courts would have jurisdiction over this matter? 2. What would be the best venue and why? 3. If a Delaware court decides that it does...
-
How might you use mass spectrometry to distinguish between the following pairs of isomers? (a) 3-Methyl-2-hexanone and 4-methyl-2-hexanone (b) 3-Heptanone and 4-heptanone (c) 2-Methylpentanal and...
-
Each of the following substances can be prepared by a nucleophilic addition reaction between an aldehyde or ketone and a nucleophile. Identify the reactants from which each was prepared. It the...
-
Describe the possible forms of audit reports for a U.S.-based client that is issuing a report for use primarily outside the United States.
-
Besides the effect on the tax rates that will apply, filing status also has an impact on the amount of the standard deduction that is allowed. A taxpayer who is unmarried or separated from their...
-
Dave borrowed $600 on January 1, 2022. The bank charged him a $8.00 service charge, and interest was $47.20. If Dave paid the $600 in 12 equal monthly payments, what was the APR? Note: Do not round...
-
You are considering an investment in Eicher Motors stock and wish to assess the firm's long - term debt - paying ability and its use of debt financing. All of the ratios can be used to assess...
-
If the future value of an ordinary, 6-year annuity is $9,100 and interest rates are 9.5 percent, what's the future value of the same annuity due? (Round your answer to 2 decimal places.)
-
Required information [The following information applies to the questions displayed below.] George and Wanda received $30,500 of Social Security benefits this year ($11,700 for George; $18,800 for...
-
Let's consider the harmonic oscillator immersed in a heat bath of temperature \(T\). For any Hermitian operator \(\hat{A}\), we can define its thermal average denoted as...
-
What are some of the possible sources of information about a company that could be used for determining the companys competitive stance?
-
Use the NH 3 molecule as an example to explain the difference between molecular geometry and electron-group geometry.
-
Provide a structural formula for the product from each of the following reactions. (a) (b) (c) (d) (e) (f) (1) LDA (2) CH,CH Br. CH,COH NaH Cl Br2 (excess), NaOH (1) LDA (3) H20 ), NaoH
-
Write a stepwise mechanism for each of the following reactions. (a) (b) (c) (d) Br Br2, CH CO H + HBr excess la NaoiH O Na t CHl + MeOH MeO CH OH, H,So OCH3
-
Would optically active ketones such as the following undergo acid-or base-catalyzed racemization? Explain your answer.
-
Subtract and simplify: 5x-6x+2-(-2-x+2x)
-
In order to value a company, you would need to forecast its future (a key word here) free cash flows. How would you approach this task? Using downloaded real data, try to make the forecast and see,...
-
share five (5) pieces of advice you would give the class about personal financial planning. Explain each. You might go back and look at the introductions in the first discussion for a general,...

Study smarter with the SolutionInn App


