Tell whether the following compounds are ortho-, meta-, orpara-di-substituted: (a) CI. CH3 (b) NO2 (c) SO3H Br
Question:
Tell whether the following compounds are ortho-, meta-, orpara-di-substituted:
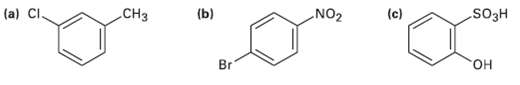
Transcribed Image Text:
(a) CI. CH3 (b) NO2 (c) SO3H Br он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
An ortho disubstituted benzene has two substituents in a 12 ...View the full answer

Answered By

Patrick Busaka
I am a result oriented and motivated person with passion for challenges because they provide me an opportunity to grow professionally.
5.00+
38+ Reviews
58+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Tell whether the following compound is chiral. trans- I,2-dimethylcyclopropane
-
The following compounds are only slightly soluble in water, but one of them is very soluble in a dilute aqueous solution of sodium hydroxide. The other is still only slightly soluble. (a) Explain...
-
The following compounds are listed in increasing order of acidity. In each case, the most acidic proton is shown in red. (a) Show the structure of the conjugate base of each acid, including any...
-
A ping pong ball is drawn at random from an urn consisting of balls numbered 4 through 9. A player wins $1.5 if the number on the ball is odd and loses $1.5 if the number is even. Let x be the amount...
-
If you were advising a North Dakota company about its selection process, would you advise it to relax its selection criteria during the oil boom? Why or why not?
-
How many stereoisomers are possible for 4-methyl-2-hexene, and how many fractions would you obtain if you distilled the mixture?
-
Explain the purpose and nature of cutoff tests of cash balances.
-
The Goodstone Tire Company produces a brand of tire called the Rain path. The annual demand at its distribution center is 12,400 tires per year. The transport and handling costs are $2600 each time a...
-
1 What are the advantages of Agile PM? What is it useful for developing new products? 1.2 Why is it difficult to apply Agile PM to large-scale projects? 1.3 Explain the concept of Minimum Viable...
-
On August 1, 2016, Stephanie Ram, a sole proprietor, started a new business, Ram Wholesale Company. The company sells refrigerators (merchandise) to various retail stores and uses the periodic...
-
Amines are converted into alkenes by a two-step process called the Hofmann elimination. SN2 reaction of the amine with an excess of CH3I in the first step yields an intermediate that undergoes E2...
-
Give IUPAC names for the followingcompounds: CH NH2 (a) Cl Br (b) (c) CH2CH2CHCH3 Br (f) CH (e) (d) CI. CH H CH-CH O2N" NO2 "C
-
Which of the following amounts paid by an employer to an employee is not subject to withholding? a. Salary b. Bonus c. Commissions d. Reimbursement of expenses under an accountable plan e. All of the...
-
Consider a sealed cylindrical container released from a height h = 3.0 m and rolling without friction on an inclined plane. How much water should be added to the container so that its final speed,...
-
Acme Soap Company manufactures two types of bars of soap, Pumice and Lavender. Data on these two products are as follows: Pumice Lavender Annual Sales volume in units 80,000 120,000 Unit Sales Price...
-
Yung Reeds, a manufacturer of saxophone, oboe, and clarinet reeds, has projected sales to be $904,000 in October, $964,000 in November, $1,060,000 in December, and $932,000 in January. Yung's sales...
-
Finished goods inventory, ending The following data is provided for Garcon Company and Pepper Company for the year ended December 31. Finished goods inventory, beginning Work in process inventory,...
-
A child is playing with a toy comprised of a massless 85-cm string attached to a small 0.04-kg ball. the child holds one end of the string and swings the ball in a vertical circle so that the ball...
-
A standard nuclear power plant generates \(3.0 \mathrm{GW}\) of thermal power from the fission of \({ }^{235} \mathrm{U}\). Experiments show that, on average, \(0.19 \mathrm{u}\) of mass is lost in...
-
A genetically engineered strain of Escherichia coli (E. coli) is used to synthesize human insulin for people suffering from type I diabetes mellitus. In the following simplified reaction scheme,...
-
Explain how you might deduce the equilibrium constant for a reaction in which you know the initial concentrations of the reactants and products and the equilibrium concentration of only one reactant...
-
Propose a mechanism for the base-catalyzed epimerization of erythrose to a mixture of erythrose and threose.
-
Show how another enediol rearrangement can move the carbonyl group from C2 in fructose to C3.
-
Show how another enediol rearrangement can move the carbonyl group from C2 in fructose to C3. Discuss.
-
An investment offers the following cash flows: $554 today, $182 one year from now, $482 in 2 years, and $793 in 3 years. If the relevant interest rate is 7% per year (an APR, with interest compounded...
-
What is the internal rate of return (IRR) for a project that costs $5,500 and is expected to generate $1,800 per year for the next four years? If the firm's required rate of return is 8 percent, what...
-
Assume B Corporation has $375,000 of Ordinary Income from its operations, $75,000 of interest received from its investments on bonds from Z firm. Also, it received $50,000 of dividends from a...

Study smarter with the SolutionInn App


