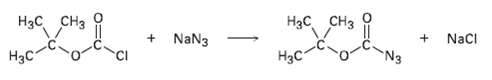
Tert-Butoxycarbonyl azide, a reagent used in protein synthesis, is prepared by treating tert-Butoxycarbonyl chloride with sodium azide.
Question:
Tert-Butoxycarbonyl azide, a reagent used in protein synthesis, is prepared by treating tert-Butoxycarbonyl chloride with sodium azide. Propose a mechanism for thisreaction.
Transcribed Image Text:
Нас, снз H3C CH3 + NaN3 NacI Нас N3 H3C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
CH33CO 0 3 addition of azide CH33CO N3 C...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Propose a mechanism that shows why p-chlorotoluene reacts with sodium hydroxide at 350 C to give a mixture of p-cresol and m-cresol.
-
Propose a mechanism for the synthesis of methyl orange.
-
A stiff, straight, horizontal wire of length 25 cm and mass 20 g is supported by electrical contacts at its ends, but is otherwise free to move vertically upward. The wire is in a uniform, horizontal...
-
Describe the advantages of face-to-face communication as opposed to interactions facilitated by technology such as telephones, e-mail, instant messaging, texting, the Web, social networking sites,...
-
A professor is interrupted independently by phone calls (at rate 1.3/h), by students with questions (at rate 0.6/h), and by colleagues (at rate 0.3/h). How many interruptions might she expect during...
-
Why does it make sense to model computer vision systems on the human vision system? Can you think of any suitable alternative models?
-
It is time to negotiate a new contract with some of Lightning Wholesale's unionized employees. The company believes in dealing fairly with its employees. Based on the current economic environment,...
-
What would be the outcome if a leader who was not performing, not communicating effectively, or building relationships were to change? What underlying causes for the leader's behavior, and what can...
-
An acoustic double-slit system (of slit separation d and slit width a) is driven by two loudspeakers as shown in Figure. By use of a variable delay line, the phase of one of the speakers may be...
-
When ethyl benzoate is heated in methanol containing a small amount of HC1, methyl benzoate is formed. Propose a mechanism for the reaction.
-
We said in Section 21.6 that mechanistic studies on ester hydrolysis have been carried out using ethyl propanoate labeled with 180 in the ether- like oxygen. Assume that 180-labcled acetic acid is...
-
What happens if two single- server systems have the same mean arrival and service rates, but the service time is constant in one and exponential in the other?
-
Sandhill manufactures mountain bikes. Its sales mix and contribution margin information per unit are as follows: Sales Mix Contribution Margin Destroyer 16% $150 Voyager 61% 57 Rebel 23% 39 It has...
-
Suppose the items below are all the items on Zex Inc.'s balance sheet in millions of Canadian dollars. Cash = $5.00 Accounts payable = $12.00 Other current liabilities = $65.00 Accounts receivable =...
-
The data to the right pertain to the operating budget of a manufacturing company. Compute the following. (a) contribution margin (b) contribution rate Sales (c) break-even point in sales dollars...
-
Insomniac records is a record label specializing in underground music artists. Each year, Insomniac records total earnings are taxed at the current corporate tax rate, and then taxed as personal...
-
Lawson Consulting had the following accounts and amounts on December 31. Cash Accounts receivable Equipment Accounts payable Common stock $ 5,000 4,500 Dividends Services revenue 6,500 Rent expense...
-
What is more plentiful on Earththe group of minerals known as feldspars or the group of minerals known as silicates?
-
In Problems, solve each system of equations. x + 2y + 3z = 5 y + 11z = 21 5y + 9z = 13
-
Determine the molecular geometry and sketch each molecule or ion using the bond conventions shown in Representing Molecular Geometries on Paper in Section 11.4. a. SF 4 b. ClF 3 c. IF 2 d. IBr 4
-
Complete the reactions given in Fig. P14.45 using knowledge or intuition developed from this or previous chapters. (a) (b) CH CH MgBr DO CH,CH CH CH2 CH 0 CH CH2-O S-O- CH2CH3 diethvl sulfate
-
Complete the reactions given in Fig. P14.45 using knowledge or intuition developed from this or previous chapters. (a) (b) CH CH MgBr DO CH,CH CH CH2 CH 0 CH CH2-O S-O- CH2CH3 diethvl sulfate
-
A student consulted a well-known compilation of reference spectra for the proton NMR spectrum of propyne and was surprised to find that this spectrum consists of a single unsplit resonance at 51.8....
-
Explained the different type of leader -Great man -trait theory -skill theory -behavior theory -Contingency theory (situational) -Transformation transactional leadership -transactional leadership...
-
What effect will a corporation's emphasis on environmental responsibility have on its financial performance in both the short term and long term? Does this contradict the notation of maximizing the...
-
Examine the emergence of technology and electronic health systems in health care since the passage of the Health Insurance Portability and Accountability Act (HIPAA). Provide an analysis of the...

Study smarter with the SolutionInn App


