Tetracaine is a substance used medicinally as a spinal anesthetic during lumbar punctures (spinal taps). (a) How
Question:
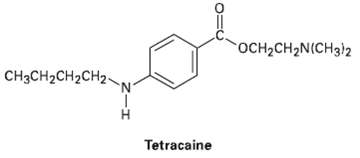
Tetracaine is a substance used medicinally as a spinal anesthetic during lumbar punctures (spinal taps).
(a) How would you prepare tetracaine from the corresponding aniline derivative, ArNH2?
(b) How would you prepare tetracaine from p-nitro benzoic acid?
(c) How would you prepare tetracaine frombenzene?
Transcribed Image Text:
OCH2CH2N(CH3)2 CHяCH2CH2CH2- Tetracaine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
CH3CH23NH 0N b 0N Tetracaine COCHCHNCH32 H Pt NCH EtOH COCHCHNCH32 COH ...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare aniline from the following starting materials? (a) Benzene (b) Benzamide (c) Toluene
-
How would you prepare o-hydroxyphenyl-acetaldehyde from phenol? More than one step is required. HO o-Hydroxyphenylacetaldehyde CH-CO
-
How would you prepare 72.5 g of an aqueous solution that is 5.00% potassium iodide, KI, by mass?
-
Coin Flips You flip a coin 100 times and get 58 heads and 42 tails. Calculate the chi-square statistic by hand showing your work, assuming the coin is fair.
-
Use one of the websites listed in Table 13.1 on page 357 to find a job opening in your target profession. If you haven't narrowed down to one career field yet, chose a business job for which you will...
-
The clutch system shown in Fig. P1025 is used to transmit torque through a 2-mm-thick oil film with μ = 0.38 Ns/m 2 between two identical 30-cm-diameter disks. When the driving shaft...
-
The Student Winner was Ulf Aslak Jensen, for the video "How People Gather: An Interactive Visualization Approach." Watch this video, and answer the following questions: (a) What data are this video...
-
After all revenue and expense account have been closed at the end of the fiscal year, Income Summary has a debit of $315,000 and a credit of $449.500. At the same date, Retained Earnings has a credit...
-
Problem 6. (a) How many quadratic residues are in the residue set Z19? (b) Determine if 4 and 9 are quadratic residues mod 19. (c) Can you solve the congruence x 8 (mod 19)? [6 points]
-
One model A1100 Kawasaki Jet Ski Personal Watercraft was sold for cash. This model retails for $9,375.00 plus 8% sales tax ($9,375.00 x 8% = $750.00). Invoice Number: G4011. Because a perpetual...
-
Cyclopentamine is an amphetamine-like central nervous system stimulant. Propose a synthesis of Cyclopentamine from materials of five carbons orless. CH3 -CH2CHNHCH3 Cyclopentamine
-
Atropine, C 17 H 23 NO 3 , is a poisonous alkaloid isolated from the leaves and roots of Atropa belladonna, the deadly nightshade. In small doses, atropine acts as a muscle relaxant; 0.5ng (nanogram,...
-
Sumitomo Chemical of Japan has one week in which to negotiate a contract to supply products to a U.S. company at a dollar price that will remain fixed for one year. What advice would you give...
-
In a more aggressive scenario (1,280,500 total assets and long-term interest rate 10% short term interest rate 7%)) with 29% using long -term financing and 71% using short-term financing, what would...
-
At the beginning of the month, the Floyd Company began when owners invested $80,000 and then during the month the company had the following transactions: Purchased equipment for $800 on account Sold...
-
In order to ensure that savings are included in the financial plan to meet Cassidy's goals, what entry must be selected in Naviplan?
-
Critical Performance Statement: Describe the purpose and importance of observation and pedagogical documentation to the profession of early childhood education and care. Apply ethical and...
-
The USFG should prioritize privacy over security. You can choose if you would rather be affirmative (saying yes they should prioritize privacy and why) or you can be negative (saying no they should...
-
What is included in cash for a business?
-
Find the velocity, acceleration, and speed of a particle with the given position function. r(t) = (t 2 , sin t - t cos t, cos t + t sin t), t > 0
-
Which factors affect the relative acidity of an oxyacid?
-
Which of the following compounds can exist as cis-trans isomers? If such isomerism is possible, draw the structures in a way that clearly illustrates the geometry. a. 3-octene b. 3-chloropropene c....
-
The mold metabolite and antibiotic mycomycin has the formula: Number the carbon chain, starting with the carbonyl carbon. a. Which multiple bonds are conjugated? b. Which multiple bonds are...
-
Write structural formulas for the following: a. 1,4-dichloro-2-pentene b. 3-hexyne c. 1,2-diethylcyclobutene d. 2-bromo-1,3-pentadiene
-
Describe and analyze issues in of subordination and privilege, oppression and resistance in their own lives and the lives of other cultural groups related to sports ?
-
Analyze each transaction. Under each category in the accounting equation, indicate whether the transaction: A. increases, B. decreases, or C. has no effect. The item (a) is provided as an example....
-
A motorist travels 70 mi while driving in a bad rainstorm. In sunny weather, the motorist drives 30 mph faster and covers 130 mi in the same amount of time. Find the speed of the motorist in the...

Study smarter with the SolutionInn App


