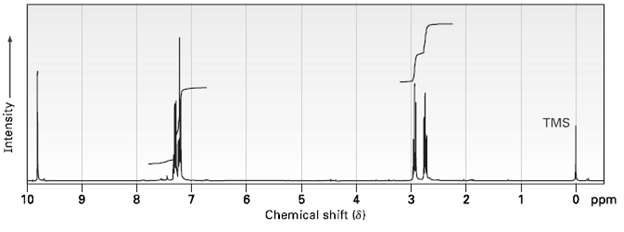
The 1 H NMR spectrum shown is that of a compound isomeric with the one in Problem
Question:
The 1H NMR spectrum shown is that of a compound isomeric with the one in Problem 19.65. This isomer has an IR absorption at 1730 cm?1. Propose a structure.
Transcribed Image Text:
TMS 10 O ppm 1. Chemical shift (8) Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
The IR absorption is that of an al...View the full answer

Answered By

Poonam Chaudhary
I have 15 month+ Teaching Experience
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1 H NMR spectrum shown is that of a compound with formula C 9 H 10 O. How many double bonds and/or rings does this compound contain? If the unknown has an IR absorption at 1690 cm ?1 , what is a...
-
The 1H NMR spectrum shown is that of 3-methyl-3-buten-l-ol. Assign all the observed resonance peaks to specific protons, and account for the splittingpatterns. TMS H H2H20 10 8. O ppm Chemical shift...
-
The 1H NMR spectrum shown in Figure 14.8 corresponds to one of the following compounds. Which compound is responsible for this spectrum? C CH CH3 CH3 CICH2 CH2C Br2CH CHBr2 6 5 3 2 0 8 (ppm)...
-
Two pulses A and B are moving in opposite directions along a taut string with a speed of 2.00 cm/s. The amplitude of A is twice the amplitude of B. The pulses are shown in Figure P18.2 at t = 0....
-
Evaluate the actions taken by the HR director in this scenario. What was done well, poorly?
-
Determine the 4-day SMA for the ten consecutive day closing prices for Wal-Mart Stores Inc listed below. $57.35, $58.61, $57.98, $58.07, $57.50 $56.97, $56.35, $56.83, $57.16, $57.18
-
What are system operational cycles?
-
Assume Ashland Community Hospital had the following supplies costs for two products used in its operating room. Standard costs for one surgery: Item X, 5 pieces at $50 each; Item Y, 10 pieces at $75...
-
Q8: Capacity Planning Cycle Time PRIMOS AMERICAN PIZZA (3 Marks) Also, PRIMO's Pizza has the process displayed below. The preparing operation occurs simultaneously. The product only needs to go...
-
1. Design an ammeter with three scales: 0-200 EA, 0-2 mA, and 0-10 mA. Use a current sensor rated at 200 EA, 100 mv. What resistance does the instrument insert in the circuit at each scale? 2. Using...
-
Compound B is isomeric with A (Problem 19.63) and shows an IR peak at 1715 cm1. The 1H NMR spectrum of B has peaks at 2.4 (1 H, septet, J = 7 Hz), 2.1 (3 H, singlet), and 1.2 (6 H, doublet, J =...
-
Propose structures for molecules that meet the following descriptions. Assume that the kinds of carbons (1, 2, 3, or 4) have been assigned by DEPT-NMR. (a) C 6 H 12 O; IR: 1715 cm 1 ; 13 C NMR: 8.0 ...
-
You are working on the Devon Inc. audit. The company is responsible for managing large-scale construction projects for its customers. During the risk assessment phase, you noted a higher risk in the...
-
In your opinion, how does financial aid contribute to student success in the for-profit and not-for-profit institutions? Is there any value in viewing the higher education industry from a business...
-
How could EMS services play a role in advancing the CCMC (Committee on the cost of medical care) guideline advocating for the extension of all basic public health services to the entire population?
-
What do aggregate demand and supply curves look like in Ethiopia (real world)? Provide me the graphical representation to back up AD and AS curves.
-
Preliminary analytical reviews are performed to obtain an understanding of the business and its environment. Analytical procedures are an important part of the audit process and are applied during...
-
What is the cost per equivalent unit for materials?
-
To determine the eigenvalues of the Hamiltonian of hydrogen we constructed the operators \(\hat{T}\) and \(\hat{S}\) from angular momentum and the Laplace-Runge-Lenz operator. The actual form of...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
Without referring to tables in the text, indicate which of the following bonds you would expect to have the greatest bond length, and give your reasons. (a) O 2 ; (b) N 2 ; (c) Br 2 ; (d) BrCl.
-
Using as starting materials 2-chloropropanoic acid, aniline, and 2-naphthol, propose a synthesis of naproanilide, a herbicide used in rice paddies in Asia: Naproanilide
-
When compound W (C15H17N) is treated with benzenesulfonyl chloride and aqueous potassium hydroxide, no apparent change occurs. Acidification of this mixture gives a clear solution. The 1H NMR...
-
Propose structures for compounds X, Y, and Z: The 1H NMR spectrum of X gives two signals, a multiplet at δ 7.3 (5H) and a singlet at δ 4.25 (2H); the 680-840-cm-1 region of...
-
Martin Company applies manufacturing overhead based on direct labor hours. Information concerning manufacturing overhead and labor for the year is as follows: Actual manufacturing overhead Estimated...
-
$ 719,000 280,000 439,000 150,400 Joyner Company's income statement for Year 2 follows: Sales Cost of goods sold Gross margin Selling and administrative expenses Net operating income Nonoperating...
-
FIFO and LIFO costs under perpetual inventory system The following units of an item were available for sale during the year: Beginning inventory 22 units at $44 Sale 18 units at $62 First purchase 18...

Study smarter with the SolutionInn App


