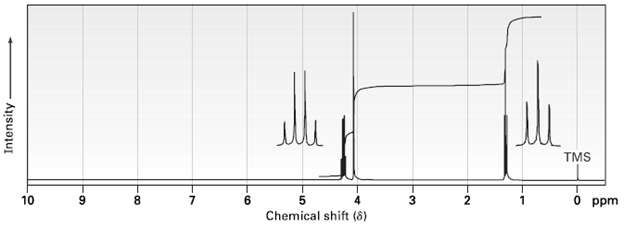
The compound whose 1 H NMR spectrum is shown has the molecular formula C 4 H 7
Question:
The compound whose 1H NMR spectrum is shown has the molecular formula C4H7O2C1 and has an infrared absorption peak at 1740 cm?1. Propose a structure.
Transcribed Image Text:
TMS 10 O ppm Chemical shift (8) Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Possible structures for C4HClO are CH3CHCOCHCl and CICHCOCHCH3 Chemical shift data can distingu...View the full answer

Answered By

Sheikh Muhammad Ibrahim
During the course of my study, I have worked as a private tutor. I have taught Maths and Physics to O'Level and A'Level students, as well as I have also taught basic engineering courses to my juniors in the university. Engineering intrigues me alot because it a world full of ideas. I have passionately taught students and this made me learn alot. Teaching algebra and basic calculus, from the very basics of it made me very patient. Therefore, I know many tricks to make your work easier for you. I believe that every student has a potential to work himself. I am just here to polish your skills. I am a bright student in my university. My juniors are always happy from me because I help in their assignments and they are never late.
4.90+
14+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The ketone whose 1H NMR spectrum is shown here was obtained as the product of an acetoacetic ester synthesis. What alkyl halide was used in the synthesis? 10 (ppm)
-
The following 1H NMR spectrum is that of an alcohol, C8H10O. Propose a structure. TMS 6. Chemical shift (8) 3 O ppm 10 8. Intensity
-
An infrared absorption spectrum of an organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this compound is more likely...
-
Estimates have been presented to Holly Farms, which is considering two environmental chambers for a project that will detail laboratory confirmations of on-line bacteria tests in chicken meat for the...
-
Some contracts, such as those between municipalities and highway construction firms, are extremely long with terms spelled out in minute detail. Others, such as those between consulting firms and...
-
Point charges of 50 nC each are located at A(1, 0, 0), B(1, 0, 0), C(0, 1, 0), and D(0,1, 0) in free space. Find the total force on the charge at A.
-
Specifically, we examine the 2004 Survey of Consumer Finances (SCF), a nationally representative sample that contains extensive information on assets, liabilities, income, and demographic...
-
Cost-Cutting Proposals Chatman Machine Shop is considering a four-year project to improve its production efficiency. Buying a new machine press for $530,000 is estimated to result in $205,000 in...
-
Required information [The following information applies to the questions displayed below.] Camille Sikorski was divorced in 2018. She currently provides a home for her 15-year-old daughter Kaly. Kaly...
-
Shelia Young started a real estate business at the beginning of January. After approval by the state for a charter to incorporate, she issued 1,000 shares of stock to herself and deposited $20,000 in...
-
Propose structures for compounds that fit the following 1H NMR data: (a) C5H10O 0.95 (6 H, doublet, J = 7 Hz) 2.10 (3 H, singlet) 2.43 (1 H, multiplet) (b) C3H5Br 2.32 (3 H, singlet) 5.35 (1 H,...
-
Propose structures for compounds that fit the following 1 H NMR data: (a) C 4 H 6 Cl 2 2.18 (3 H, singlet) 4.16 (2 H, doublet, J = 7 Hz) 5.71 (1 H, triplet, I = 7 Hz) (b) C 10 H 14 1.30 (9 H,...
-
In several exercises in this chapter, we considered the study of one week of a college football season, during which the population of 53 FBS games had a mean spread (winning score minus losing...
-
Do you think investors understand what happens to individual mortgages once they are bundled and securitized?
-
Why do some companies emphasize career planning with current employees?
-
In the low-urgency scenarios, which actions were the most effective in achieving your desired outcomes and why? What about the high-urgency scenarios? Why is it more difficult to lead change in a...
-
IBM is selling a $1,000 par value bond that has a coupon rate of 4% that pays interest annually and has 10 years remaining to maturity. Assuming you require a yield to maturity of 8 percent, what is...
-
What is one step an employee can take to maintain work-life balance by setting clear communication boundaries?
-
A machine makes pipe fittings to have a mean length of approximately \(2 \mathrm{in}\). and the standard deviation of the lengths must be less than \(0.04 \mathrm{in}\). To make sure the machine is...
-
The graph of the sequence whose general term is an = n - 1 is which of the following? [8.1] A. B. TITTT 3-2-1 23.45 2.3.4
-
Scuba divers breathing air at increased pressure can suffer from nitrogen narcosisa condition resembling drunkennesswhen the partial pressure of nitrogen exceeds about 4 atm. What property of...
-
Predict the products of the following proposed Diels-Alder reactions. (a) (b) (c) (d) (e) (f) CHO C-C-IC-C CN NC CN O + OCHs CN CH,O CN
-
What dienes and dienophiles would react to give the following Diels-Alder products? (a) (b) (c) (d) (e) (f) C-CH CH3O CN C OCH C--OCH, CN CN CH,O CN H O
-
Predict the major product for each proposed Diels-Alder reaction. Include stereochemistry where appropriate. (a) (b) (c) Ph 0 Ph
-
6x+17x-3 Evaluate the following limit: lim xo 5x2+14x-3
-
Find all the critical numbers of y=x-2x-3, then determine the local minimum and maximum points by using a graph.
-
At September 3 0 , the end of Beijing Companys third quarter, the following stockholdersequity accounts are reported. Common stock, $ 1 0 par value $ 4 8 0 , 0 0 0 Paid - in capital in excess of par...

Study smarter with the SolutionInn App


