The dehydration of citrate to yield cis-aconitate, a step in the citric acid cycle, involves the pro-R
Question:
The dehydration of citrate to yield cis-aconitate, a step in the citric acid cycle, involves the pro-R ?arm? of citrate rather than the pro-S arm. W1ich of the following two products is formed?
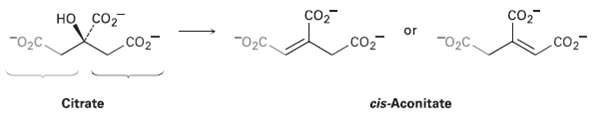
Transcribed Image Text:
но со "02C. Co2 Co2 or "02C. CO2 Co2 .co2 Citrate cis-Aconitate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 52% (17 reviews)
If you perform the replacement test to ...View the full answer

Answered By

Nimlord Kingori
2023 is my 7th year in academic writing, I have grown to be that tutor who will help raise your grade and better your GPA. At a fraction of the cost on other sites, I will work on your assignment by taking it as mine. I give it all the attention it deserves and ensures you get the grade that I promise. I am well versed in business-related subjects, information technology, Nursing, history, poetry, and statistics. Some software's that I have access to are SPSS and NVIVO. I kindly encourage you to try me; I may be all that you have been seeking, thank you.
4.90+
360+ Reviews
1070+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The first step in the citric acid cycle is reaction of oxaloacetate with acetyl CoA to give citrate. Propose a mechanism, using acid or base catalysis asneeded. "O2C. 02C H .co2 CO SCOA Citrate...
-
(a) Explain which of the following two products is formed when the reactant alkyl chloride reacts with sodium acetate in DMSO. (b) Explain which of the following two product alcohols is formed when...
-
Which of the substances in the citric acid cycle are tricarhoxylic acids, thus giving the cycle its alternative name?
-
Being able to identify an industry to use for benchmarking yourfirm's results with similar companies is frequently not easy. Choose a type of business and go to www.naics.com. This Web site allows...
-
A railroad diesel engine weighs four times as much as a freight car. If the diesel engine coasts at 5 km/h into a freight car initially at rest, show that the speed of the coupled engine and car is 4...
-
Derive the following formula for the distance e from the centerline of the wall to the shear center S for the C-section of constant thickness shown in the figure: Also, check the formula for the...
-
What are the two models of quality of service and how are they different?
-
Matilda Crone owns and operates a public relations firm called Dance Fever. The following amounts summarize her business on August 31, 2012: During September 2012, the business completed the...
-
This assignment relates to the following Course Learning Requirements: CLR1 - Assess and apply both the internal and external environments of an organization to an employee compensation system CLR2 -...
-
On January 1, 2014, Prince Corporation acquired 70% of the 100,000 outstanding voting shares of Song Limited for a cash consideration of $1,015,000. On that date, shares of Song Limited were trading...
-
One of the steps in fat metabolism is the hydration of crotonate to yield 3-hydroxybutyratc. The reaction occurs by addition of OH to the Si face at C3, followed by Protonation at C2, also from the...
-
The first step in the metabolism of glycerol formed by digestion of fats is phosphorylation of the pro-R CH 2 OH group by reaction with All to give the corresponding glycerol phosphate. Show the...
-
Greene Company sold its 100% WI in a proved property for $700,000 and retained an ORI. Greenes net cost basis in the property was $450,000. The fair market value of the entire original working...
-
Show that argz -arg|z And arg(1/z) = -argz
-
Insert n or U to make the statement true. (8,12,16,20) (1,8,9,12,15) = (8,12)
-
Put the equation y = x x - Answer: y = +63 16x + 63 into the form y = (xh)+k:
-
Factor the following expression. 8x-4x+12x 4x
-
Evaluate -5 (-5)-7.
-
What principle that relates events depends on the arrow of time? State this principle, and briefly explain what it means.
-
The purpose of this case is to come up with a contingency plan[s] in order to sustain the program Move With Me, a program that serves thousands of community members throughout Lower Manhattan. The...
-
People sometimes use sodium bicarbonate as an antacid to neutralize excess hydrochloric acid in an upset stomach. What mass of hydrochloric acid (in grams) can 2.5 g of sodium bicarbonate neutralize?
-
The stability of a DNA double helix can be measured by its melting temperature, Tm, defined as the temperature at which the helix is 50% dissociated into individual chains. Which of the following...
-
When DNA is treated with 0.5 M NaOH at 25C, no reaction takes place, but when RNA is subjected to the same conditions, it is rapidly cleaved into mononucleotide 2- and 3-phosphates. Explain.
-
Indicate whether you would expect imidazole to have high or low water solubility, and why.
-
1. Write a Java program that will prompt the user for a number and print out a square with those dimensions. For example, if the user enters 5, return the following: * * * ** ** * * * * * * * * * * *...
-
2. Vector multiplication or dot product is performed by multiplying corresponding elements and summing the products. It can be represented mathematically as oa; *bi where a and b are vectors of...
-
The following data was collected from an experiment testing the hypothesis: The density of cream whipped for 5 minutes increases at higher altitudes. What would be an acceptable interpretation of...
Practice Problems Workbook Engineering Mechanics Statics 13th Edition - ISBN: 0132915596 - Free Book

Study smarter with the SolutionInn App


