The distribution coefficient for extraction of a metal complex from aqueous to organic solvents is D =
Question:
The distribution coefficient for extraction of a metal complex from aqueous to organic solvents is D = [total metal]org/[total metal]aq. Give physical reasons why β and Ka appear in the numerator of Equation 22-13, but KL and [H+]aq appear in the denominator.
Equation 22-13
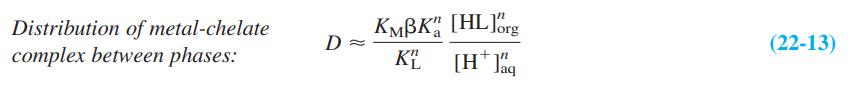
The word "distribution" has several meanings in the financial world, most of them pertaining to the payment of assets from a fund, account, or individual security to an investor or beneficiary. Retirement account distributions are among the most...
Transcribed Image Text:
KMBK", [HL]brg Distribution of metal-chelate complex between phases: (22-13) KE [H* J%q
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The form that is extracted into organic solvent is MLW The formation of ML n is f...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
For the extraction of Cu 2+ by dithizone in CCl 4 , KL= 1.1 10 4 , K M = 7 10 4 , K a = 3 10 - 5 , = 5 10 22 , and n = 2. (a) Calculate the distribution coefficient for extraction of 0.1 M Cu 2+...
-
An aqueous acetic-acid solution containing 6.0 moles of acid per liter is to be extracted in the laboratory with chloroform at 25oC to recover the acid (B) from chloroform-insoluble impurities...
-
Consider the extraction of 100.0 mL of M2+ (aq) by 2.0 mL of 1 10-5 M dithizone in CHCl3, for which KL = 1.1 104, KM = 7 104, Ka = 3 10-5, = 5 1018, and n = 2. (a) Derive an expression for the...
-
Analyze how you will use the challenge the process and enable others to act practices to improve the three leadership areas that you selected in module one. ? select one leadership theory or approach...
-
At May 31, Suarez Company has net sales of $330,000 and cost of goods available for sale of $230,000. Compute the estimated cost of the ending inventory , assuming the gross profit rate is 35%.
-
Business success is often tied to effectively managed strategies. Using the Internet, study Starbuck's current performance. Based on analysis, do you judge Starbucks to be a success? Why or why not?...
-
Find the stiffness and mass matrices of the beam supported on springs as shown in Fig. 12.19. Model the beam using one finite element. Assume the material of the beam as steel with a Young's modulus...
-
The three-station work cell illustrated in figure S7.7, has a product that must go through one of the two machines at station 1 (they are parallel) before proceeding to station 2. (a) What is the...
-
Recommend 3 strategies to evaluate the business impact of change and sustain change efforts through performance management. Recommend 4 best practices for ensuring the application of new skills and...
-
Write the augmented matrix associated with each system of linear equation? 1. 2. Each matrix is an augmented matrix used in the solution of a system of linear equations in x, y, and z. What is the...
-
Consider two Gaussian peaks with relative areas of 4:1. Construct a set of graphs to show the overlapping peaks if the resolution is 0.5, 1, or 2.
-
Give a physical interpretation of Equations 22-6 and 22-7 in terms of the fractional composition equations for a monoprotic acid discussed in Section 9-5
-
End A of the bar AB in Fig. P11.53 rests on a frictionless horizontal surface, and end B is hinged. A horizontal force F of magnitude 160 N is exerted on end A. You can ignore the weight of the bar....
-
2. Ahmed has OMR 5,000 to invest for three years period, He has five investment alternatives. What will be the amount after three years from the flowing alternatives? i. Bank deposit at 5% ii. Bond...
-
2. Draw the demand curve for an environmental service, for example carbon sequestration (process of capture and long-term storage of atmospheric carbon-dioxide) from tree planting and discuss what...
-
Suppose the federal government imposes a tariff of $1 on each million bagels imported. Re-draw the demand and supply curves, along with the world supply of bagels with the tariff. Calculate the...
-
Answer the following questions based off of the graphs above, which depict a perfectly competitive industry and firm: If the industry or market is in equilibrium at point C, where will the firm...
-
Utilize the graph above, which illustrates average fixed costs, average variable costs, average total costs, and marginal costs of production for a firm in a perfectly competitive market, to answer...
-
When the electric field is at its maximum value, the magnetic energy of a cavity is (a) At its maximum value (b) At 2 of its maximum value (c) At 1/2 of Its maximum value (d) At 1/2 of its maximum...
-
What mass of KBr (in grams) should you use to make 350.0 mL of a 1.30 M KBr solution?
-
Bone consists of the protein collagen and the mineral hydroxyapatite, Ca 10 (PO 4 ) 6 (OH) 2 . The Pb content of archaeological human skeletons measured by graphite furnace atomic absorption sheds...
-
Why is an internal standard most appropriate for quantitative analysis when unavoidable sample losses are expected during sample preparation?
-
Explain why X-ray fluorescence is observed when matter absorbs X-rays of sufficient energy. Why does each element have a unique X-ray signature?
-
If you were to enter the banking industry, you might find yourself approving or not approving loans. The following is a good example of a common event you could encounter. Company X is looking for...
-
You see a two year bond with annual coupon rate of 8% that has these flows. Years ahead Cashflows 0 ?? 0.25 0 0.5 0 0.75 1 1.25 1.5 1.75 2 8 0 0 0 108 You will be asked to calculate its price at...
-
Use synthetic division and the Remainder Theorem to evaluate P(c). P(x) = 6x + 5x +9, c = 11/1 P(1) =

Study smarter with the SolutionInn App


