The experiment in Figure 16-8 required 5.32 mA for 964 s for complete reaction of a 5.00
Question:
The experiment in Figure 16-8 required 5.32 mA for 964 s for complete reaction of a 5.00 mL aliquot of unknown cyclohexene solution.
(a) How many moles of electrons passed through the cell?
(b) How many moles of cyclohexene reacted?
(c) What was the molarity of cyclohexene in the unknown?
Figure 16-8
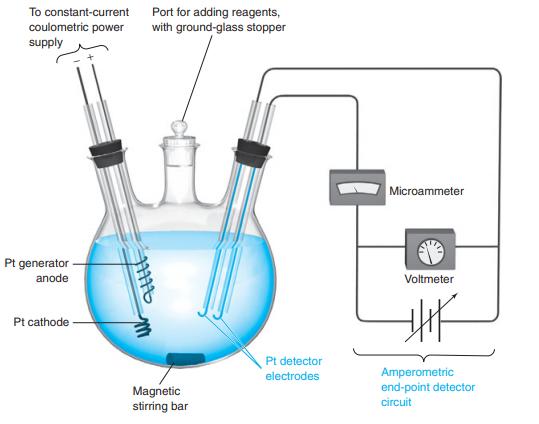
Transcribed Image Text:
To constant-current coulometric power supply Port for adding reagents, with ground-glass stopper Microammeter Pt generator anode Voltmeter Pt cathode Pt detector Amperometric end-point detector electrodes Magnetic stirring bar circuit
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a mole b One mol e reacts with mol Br 2 w...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
The active ingredients in an antacid tablet contained only calcium carbonate and magnesium carbonate. Complete reaction of a sample of the active ingredients required 41.33 mL of 0.08750 M...
-
Explain how the amperometric end-point detector in Figure 16-8 operates. Figure 16-8 To constant-current coulometric power supply Port for adding reagents, with ground-glass stopper Microammeter Pt...
-
Does the MO energy diagram of cyclooctatetraene (Figure 16-8) appear to be a particularly stable or unstable configuration? Explain. In Figure 16.8 nonbonding line cyclobutadiene cyclooctatetraene...
-
Modify Lookup to make a program LookupAndPut that allows put operations to be specified on standard input. Use the convention that a plus sign indicates that the next two strings typed are the...
-
At the beginning of 2012, Mazzaro Company acquired equipment costing $120,000. It was estimated that this equipment would have a useful life of 6 years and a salvage value of $12,000 at that time....
-
Land improvements As of December 31 2022 : Beginning balance - P5,000,000 Estimated useful life - 10 years Depreciation method - 150% declining balance method Accumulated Depreciation - P2,781,473 a....
-
How are the mean value and variance of a random variable defined?
-
The Hospital for Ending Long-term Problems (HELP) had the following financial events during the year: 1. HELP collected $ 250,000 in cash that it had billed to the federal government under Medicare....
-
What are your thoughts/reflections on the reading? Do you agree/disagree? Edwin Not Every Student Should Go to College. And That's OK By Michael B. Hom & Bob Moesta Forty years ago 32 percent of...
-
Two laptop manufacturing companies A and B are fierce rivals. A's laptop have a mean life of 4.9 years with a standard deviation of 1.2 years while B's laptop have a mean life of 4 years with a S.D...
-
The sensitivity of a coulometer is governed by the delivery of its minimum current for its minimum time. Suppose that 5 mA can be delivered for 0.1 s. (a) How many moles of electrons are delivered by...
-
H 2 S(aq) can be analyzed by titration with coulometrically generated I 2 . H 2 S + I 2 + S(s) + 2H + + 2I - To 50.00 mL of sample were added 4 g of KI. Electrolysis required 812 s at 52.6 mA....
-
Write a GUI application that lets the user enter a file name in the text field and press the Enter key to display its binary representation in a text area. The user can also modify the binary code...
-
A Gallup Poll asked a sample of Canadian adults if they thought the law should allow doctors to end the life of a patient who is in great pain and near death if the patient makes a request in...
-
Applied Manufacturing, Inc. (AM) is a custom metal fabrication corporation providing a wide range of goods and services to its customers. For decades, AM has been a parts manufacturer and supplier to...
-
Referring to the forward converter shown below, (a) Calculate the output voltage (b) Plot the following waveforms: (i) v pri, (ii)i, (iii)iin. The magnetizing inductance referred to the primary side...
-
Assume that the company produces and sells 98,000 units during the year at a selling price of $9.00 per unit. Prepare a contribution format income statement for the year. Note: Round your...
-
3. A uniform magnetic field makes an angle of 30 with the axis of a circular coil that has 300 turns and a radius equal to 4 cm. The magnitude of the magnetic field increases at a rate of 85 T/s,...
-
On January 31,Cardston Company had the following payroll liability accounts in its ledger In February, the following transactions occurred: Feb. 4 Sent a cheque to the union treasurer for union dues....
-
Imagine that your best friend knows you are taking a psychology course and wonders what psychology is all about. How would you define psychology for your friend? Write an essay on the discipline of...
-
Antibonding molecular orbitals can be used to make bonds to other atoms in a molecule. For example, metal atoms can use appropriate d orbitals to overlap with the *2p orbitals of the carbon monoxide...
-
Methyl isocyanate, CH3NCO, was made infamous in 1984 when an accidental leakage of this compound from a storage tank in Bhopal, India, resulted in the deaths of about 3,800 people and severe and...
-
(a) Methane (CH4) and the perchlorate ion are both described as tetrahedral. What does this indicate about their bond angles? (b) The NH3 molecule is trigonal pyramidal, while BF3 is trigonal planar....
-
The imposition of a ceiling on the length of time a criminal case should take to get to trial is a multifaceted issue, balancing the defendant's right to a timely trial with the complexities of...
-
Discuss three changes written into the Constitution in 1789 that changed the government from what it was under the ArticIes of Confederation?
-
Assume South Park is initially closed to trade. Domestic supply and demand for winter hats is given by the following equations: D = 60 - 4P, S = 2P - 6. a. Suppose South Park opens up to trade and...

Study smarter with the SolutionInn App


