The following are not listed as design variables for the distillation unit operations in Table 5.4: (a)
Question:
The following are not listed as design variables for the distillation unit operations in Table 5.4:
(a) Condenser heat duty
(b) Stage temperature
(c) Intermediate-stage vapor rate
(d) Reboiler heat load
Under what conditions might these become design variables? If so, which variables listed in Table 5.4 would youeliminate?
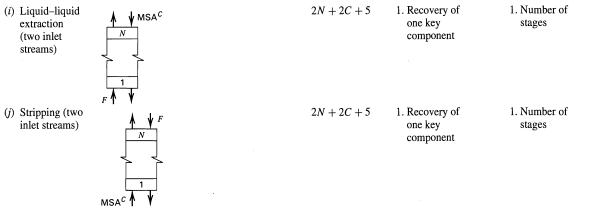
Transcribed Image Text:
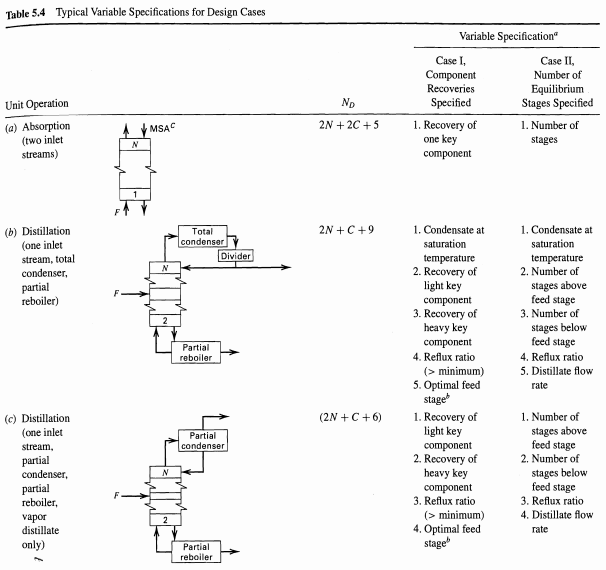
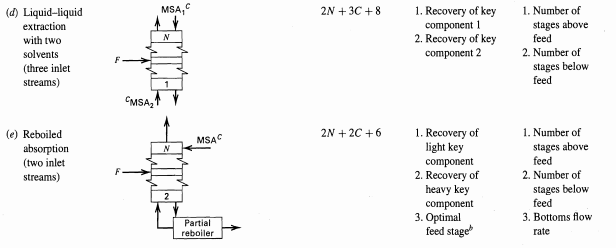
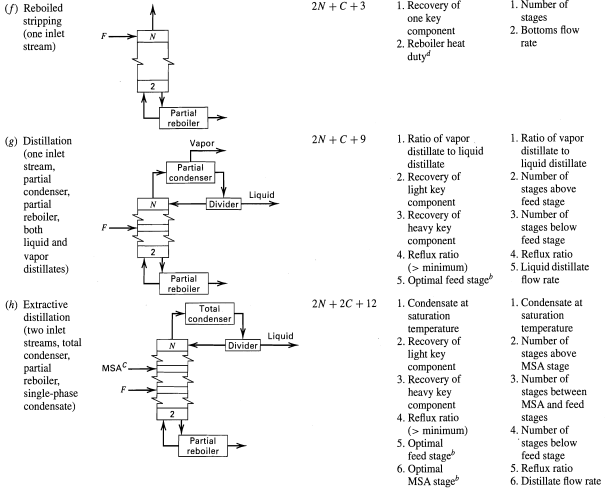
Typical Variable Specifications for Design Cases Table 5.4 Variable Specification Case I, Component Recoveries Case II, Number of Equilibrium Stages Specified Specified No Unit Operation 1. Recovery of 1. Number of (a) Absorption (two inlet streams) 2N + 2C +5 one key component stages 1. Condensate at saturation temperature 2. Recovery of light key component 3. Recovery of heavy key component 2N +C+9 1. Condensate at saturation (b) Distillation (one inlet stream, total condenser, partial reboiler) Total condenser Divider temperature 2. Number of stages above feed stage 3. Number of 2 stages below feed stage Partial 4. Reflux ratio 4. Reflux ratio reboiler 5. Distillate flow (> minimum) 5. Optimal feed stage 1. Recovery of light key rate (2N +C + 6) 1. Number of stages above feed stage (c) Distillation (one inlet Partial condenser component 2. Recovery of heavy key stream, partial condenser, partial reboiler, 2. Number of stages below feed stage 3. Reflux ratio component 3. Reflux ratio (> minimum) 4. Optimal feed stage 4. Distillate flow vapor distillate rate only) Partial reboiler
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a Condenser heat duty might be substituted for the reflux rati...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Verify ND for the following unit operations in Table 5.4: (b), (c), and (g). How would ND change if two feeds were used instead ofone? 2N +C+9 I. Condensate at 1. Condensate at (b) Distillation (one...
-
Show directly from the definition that the following are not Cauchy sequences. (a) ((-1)n, (b) n + (-1)n/n), (c) (ln n)
-
Show that each of the following are not valid rules about FDs by giving example relations that satisfy the given FDs (following the if) but not the FD that allegedly follows (after the then). a) If A...
-
Write a method: that displays the prompt string, reads an integer, and tests whether it is between the minimum and maxi mum. If not, print an error message and repeat reading the input. Add the...
-
The following information (in U.S. millions) is available for Limited Brands, Inc., the parent company of Victoria's Secret, La Senza, and Bath & Body Works, among other companies: (a) Calculate the...
-
The partnership of Par and Boo was formed and commenced operations on March 1, 2016, with Par contributing $30,000 cash and Boo investing cash of $10,000 and equipment with an agreed-upon valuation...
-
The combustion of ethanol goes according to the following equation: \[ \mathrm{C}_{2} \mathrm{H}_{5} \mathrm{OH}+\ldots \mathrm{O}_{2} ightarrow \ldots \mathrm{CO}_{2}+\ldots \mathrm{H}_{2}...
-
At December 31, 2016, Finzelberg Company had a credit balance of $15,000 in Allowance for Doubtful Accounts. During 2017, Finzelberg wrote off accounts totaling $11,000. One of those accounts...
-
On January 1, 2020, Hull Corp. paid $ 516,000 for 24% (56,000 shares) of Oliver Co outstanding common stock. Hull uses the equity method. The investment balance at the end of 2020 was $672,000. On...
-
Cherry Cotta makes custom ordered clay pots for residential gardens. Below is cost information regarding its latest job. a. Materials were purchased on account. $18,996 purchased b. A materials...
-
Verify ND for unit operation (h) in Table 5.4. How would ND change if a liquid side stream was added to a stage that was located between the feed stage and stage2? 2N + 20 + 12 1. Condensate at 1....
-
Show for distillation that, if a total condenser is replaced by a partial condenser, the number of degrees of freedom is reduced by 3, provided that the distillate is removed solely as a vapor.
-
Integrate the hydrostatic relation by assuming that the isentropic bulk modulus, B = (p/)s, is constant. Apply your result to the Mariana Trench, Prob. 2.7.
-
In what month and year did Adelphia file for bankruptcy?
-
Discuss the reasons for this phenomenon and explain the probable reasons why no one who knew about the fraud came forward to stop it?
-
What event triggered the end of the oil-for-food program?
-
What was the name of the former U.N. Secretary General Boutros Boutros-Ghalis brother-in-law who was indicted related to the oil-for-food scandal?
-
Is Mickey Monus now out of jail?
-
Which of the following is not a model organism? a. Mus musculus (laboratory mouse) b. Escherichia coli (a bacterium) c. Saccharomyces cerevisiae (a yeast) d. Sciurus carolinensis (gray squirrel)
-
In Problem use absolute value on a graphing calculator to find the area between the curve and the x axis over the given interval. Find answers to two decimal places. y = x 3 ln x; 0.1 x 3.1
-
What role does the HR function play in facilitating career development?
-
A 10 wt% aqueous solution of NaOH at 100F and a flow rate of 30,000 lb/h is to be concentrated to 50 wt% by evaporation using saturated steam at 115 psia. (a) If a single-effect evaporator is used...
-
Fifty thousand lb/h of a 20 wt% aqueous solution of NaOH at 120F is to be fed to an evaporator operating at 3.7 psia, where the solution is concentrated to 40 wt% NaOH. The heating medium is...
-
A desublimation unit of the heat-exchanger type is to be sized for the recovery of 200 kg/h of benzoic acid (BA) from a gas stream containing 0.8 mol% BA and 99.2 mol% N 2 . The gas enters the unit...
-
[3] 4. In 1995, Mary bought an antique chair for $55. It is estimated that the chair will increase in value by 1.8%/year. a) Find the current value of the chair. 121 [2] b) In what year will the...
-
Outback Outfitters sells recreational equipment. One of the company s products, a small camp stove, sells for $ 1 4 0 per unit. Variable expenses are $ 9 8 per stove, and fixed expenses associated...
-
Rowland & Sons Air Transport Service, Incorporated, has been in operation for three years. The following transactions occurred in February: February 1 Paid $ 5 5 5 to rent equipment that was used and...

Study smarter with the SolutionInn App


