The following input-output pairs have been observed during the operation of a linear system: Can you draw
Question:
The following input-output pairs have been observed during the operation of a linear system: Can you draw any conclusions about the time invariance of this system?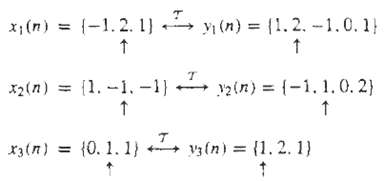
Transcribed Image Text:
x;(n) = (-1.2. 1) → y (n) = (1.2. -1.0.1} 2(n) = {-1, 1. 0.2) x2(n) = {1. -1.-1) (1. 2. 1) r3(n) = {0. 1. 1) vy(n)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (19 reviews)
Since x 1 n x 2 n n and the system is li...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Digital Signal Processing
ISBN: ?978-0133737622
3rd Edition
Authors: Jonh G. Proakis, Dimitris G.Manolakis
Question Posted:
Students also viewed these Telecommunication Engineering questions
-
The following input-output pairs have been observed during the operation of various system, determine their frequency response if each of the above system is LTI. 7, (a) xin) = (" v(n) = (})" y(n) =...
-
Give an example of a linear system that has an equilibrium solution (x(t),y(t)) such that x(0) = -1 and y(t) = 2x(t) for all t. Give an example of a linear system that has an straight line solution...
-
Suppose the coefficient matrix of a linear system of four equations in four variables has a pivot in each column. Explain why the system has a unique solution.
-
An average of three small businesses go bankrupt each month. What is the probability that five small businesses will go bankrupt in a certain month?
-
Don's Lumber Company on the Hudson River features a large selection of materials for flooring, decks, moldings, windows, siding, and roofing. The prices of lumber and other building materials are...
-
ELS corporation is about to begin its sixth year of existence. Assume that ELS reported gross receipts for each of its first five years of existence for scenarios A, B, and C as follows: Year of...
-
Pick any three of the sources of prospects discussed in the chapter and pick a product or service you like. Develop several ideas for how you would use each source to locate leads for the product or...
-
Easy-Tech Software Corporation is evaluating the production of a new software product to compete with the popular word processing software currently available. Annual fixed costs of producing the...
-
Assume that P = Tshs 1 0 0 , 0 0 0 , i = 1 2 % , and n = 3 years. Find FV when interest is compounded: ( a ) annually
-
Annie and Eddie Pennywise decide that they don't need a lawyer to represent them when they purchase their first home in Massachusetts. They read up on closings through Readers Digest and other...
-
The following input-output pairs have been observed during the operation of a time-invariant system: Can you draw any conclusion regarding the linearity of the system? What is the impulse response of...
-
The only available information about a system consist of N input-output pairs, of signals yi(n) = T[xi(n)], I = 1,2,..,N (a) What is the class of input signals for which we can determine the output,...
-
Using the given sample information, test the following hypotheses. a. H0: p1 p2 = 0.......Ha: p1 p2 0 Sample 1 .... Sample 2 n1 = 368 ... n2 = 405 x1= 175 ... x2 = 182 Let = .05. x is the number...
-
It requires 1 1 . 5 mL of a 0 . 5 0 0 M solution of KOH to titrate a 8 . 0 5 mL sample of HNO 3 . Determine the concentration of the HNO 3 solution.
-
At a given temperature, the elementary reaction AB, in the forward direction, is first order in A with a rate constant of 0.0260 s. The reverse reaction is first order in B and the rate constant is...
-
How much heat is required to heat 1 0 . 0 g of ice at - 5 . 0 0 oC to become liquid water at + 7 . 0 0 oC ? In this temperature range, the heat capacity of H O ( s ) is 3 7 . 7 J / mol * K , and the...
-
The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: = E Ryn 2 In this equation Ry stands for the Rydberg energy, and n stands for the principal quantum number of...
-
Draw the major product of the following SN2 reaction. CH3 -Br -CH3 Save Close OscerSketch Clear OscerSketch OSCER SKETCH CH B NOF Cl Br I Si P S Li Na * 1 + - ~ ++ XX CH3 -CH3 Br OH CH3
-
Med Max buys surgical supplies from a variety of manufacturers and then resells and delivers these supplies to dozens of hospitals. In the face of declining profits, Med Max decided to implement an...
-
Evaluate each logarithm to four decimal places. log 0.257
-
From the observation that 0.0500 M vinylacetic acid has a freezing point of -0.096 C, determine K a for this acid. CH2=CHCH,CO,H+H,O
-
In Section 2.9.6, it was argued intuitively that Y(e j? ) = H(e j? ) X(e j? ), When Y(e j? ), H(e j? ), and X(e j? ) are, respectively, the Fourier transforms of the output y [n], impulse response...
-
By applying the Fourier synthesis equation (Eq. (2.133)) to Eq. (2.172) and using Theorem 3 in Table 2.2, demonstrate the validity of the modulation theorem (Theorem 7, Table 2.2). FOURIER TRANSFORM...
-
Let x [n] and y [n] denote complex sequences and X(e j? ) and Y(e j? ) their respective Fourier transforms. (a) By using the convolution theorem (Theorem 6 in Table 2.2) and appropriate properties...
-
Explain why leasing is an option for a company expansion. include, what leasing is and how it will benefit the company in it's expanding efforts. Also, how is capital or operating leasing recorded on...
-
Discuss the following statement: " A head of state signs a treaty on behalf of his country in excess of authority of his country, such treaty shal be void for inconsistency with domestic law of the...
-
A company is looking at new equipment with an installed cost of $415,329. This cost will be depreciated straight-line to zero over the project's 5-year life, at the end of which the equipment can be...

Study smarter with the SolutionInn App


