The following reaction involves an intra molecular Michael reaction followed by an intra molecular aldol reaction. Write
Question:
The following reaction involves an intra molecular Michael reaction followed by an intra molecular aldol reaction. Write both steps, and show theirmechanisms.
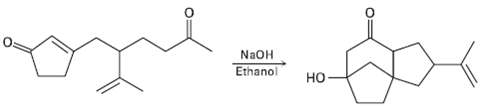
Transcribed Image Text:
NaOH Ethanol но
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
This problem becomes easier if ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
The following reaction involves an intra molecular aldol reaction followed by a ret ro aldol-like reaction. Write both steps, and show theirmechanisms. O2Et CO2Et Na* "OEt Ethanol
-
The following reaction involves two successive intra molecular Michael reactions. Write both steps, and show theirmechanisms. Nat "OEt Ethanol
-
The following reaction involves a starting material with a double bond and a hydroxyl group, yet its mechanism resembles a pinacol rearrangement. Propose a mechanism, and point out the part of your...
-
Limits from graph In this problem we evaluate limits from the graph. Consider the graph of f given in [Figure 1]. Evaluate each of the following limits (or explain why if the limit does not exist)....
-
Advertising has been likened to offering a performance bond, where the seller must relinquish the bond if certain performance goals are not met. What is the connection between advertising and bonding.
-
Chlorine reacts with oxygen to form Cl2O7. (a) What is the name of this product (see Table 2.6)? (b) Write a balanced equation for the formation of Cl2O7 (l) from the elements. (c) Under usual...
-
What are three general types of business value drivers?
-
Effect of LIFO on financial statements over several periods. Harmon Corporation commenced operations on January 1, 2008. It uses a LIFO cost-flow assumption. Its purchases and sales for the first...
-
What type of capacity planning tools would you use to ensure that your IT workplace is operating at optimal efficiency?
-
Ruth Jones, a robust 50-year-old insurance adjuster living in the northern suburbs of Chicago, has been diagnosed by a University of Illinois cardiologist as having a defective heart valve. Although...
-
The following reaction involves a hydrolysis followed by an intra molecular nucleophilic acyl substitution reaction. Write both steps, and show theirmechanisms. CH CH H30* CHCH CH2CO2H
-
The following reaction involves a conjugate addition reaction followed by an intra molecular Claisen condensation. Write both steps, and show theirmechanisms. CO2CH3 (CH3i>CuLi -CEC-CO2CH3 H
-
Compute trend percents for the following accounts, using 2007 as the base year. State whether the situation as revealed by the trends appears to be favorable or unfavorable for eachaccount. 2011 2010...
-
1) Outline why interest rates differ across borrowers, across different countries and over time. 2) Explain the various theories explaining the shape of the yield curve at any point in time and the...
-
Sundance Systems has the following transactions during July. July 5 Purchases 48 LCD televisions on account from Red River Supplies for $2,900 each, terms 3/10, n/30. July 8 Returns to Red River...
-
A Comparative Analysis of Accounting Systems in Malaysia and Pakistan Introduction: You are a financial analyst working for a multinational corporation planning to expand its operations into either...
-
The annual interest rate in the U.K. is 0. The interest rate in Canada is 1 percent. The current spot exchange rate is 1 pound = 2 CAD. For covered interest parity to hold, the 1-year forward...
-
Organizations are responsible for ensuring that their AIS applications comply with maintaining internal controls, such as segregation of duties. Technology advances such as robotic processing...
-
A block of inertia \(m\) is attached to a block of inertia \(2 m\) by a light string draped over a uniform disk of inertia \(3 m\) and radius \(R\) that can rotate on a horizontal axle (Figure...
-
Trade credit from suppliers is a very costly source of funds when discounts are lost. Explain why many firms rely on this source of funds to finance their temporary working capital.
-
Explain the difference between endothermic reactions and exothermic reactions with respect to the bond energies of the bonds broken and formed.
-
Magnesium monoperoxyphthalate (MMPP) epoxidizes alkenes much like MCPBA. MMPP is more stable, however, and it may be safer to use for large-scale and industrial reactions. Propose a mechanism for the...
-
Predict the major products of the following reactions. (a) Cis-hex-2-ene + MCPBA in chloroform (b) Trans-hex-3-ene + peroxyacetic acid (CH3CO3H) in wat (c) 1-methylcyclohexene + MMPP in ethanol (d)...
-
When 1,2-epoxycyclohexane (cyclohexene oxide) is treated with anhydrous HCl in methanol, the principal product is trans-2-methoxycyclohexanol. Propose a mechanism to account for the formation of this...
-
Research: What is a sovereign nation, how many sovereign nations are recognized as members by the United Nations and how many sovereign nations are recognized by the US State Department. Compare the...
-
Marigold Corp. purchases a patent for $198,000 on January 2, 2022. Its estimated useful life is 18 years. (a) Prepare the journal entry to record amortization expense for the first year. (Credit...
-
With an interest of $ 2 4 . 3 1 and a principal of $ 3 , 5 0 0 for 1 0 0 days, using the ordinary interest method, the rate is ?

Study smarter with the SolutionInn App


