The following reaction is an example of a [2, 3] sigma tropic rearrangement. Would you expect the
Question:
The following reaction is an example of a [2, 3] sigma tropic rearrangement. Would you expect the reaction to be suprafacial or antarafacial?Explain.
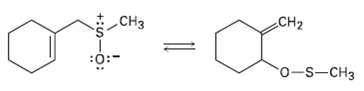
Transcribed Image Text:
CH2 :0:- о-s-CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 52% (17 reviews)
0 CH3 T 3 CH3 CH 0SCH3 Th...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Suppose that the aggregate demand for a depletable resource is given by the following inverse demand function: P= 100 (Q+S) where P is the market price, Q is the amount of depletable resource, and S...
-
Would you expect cartel formation to be more likely in industries composed of a few firms or in those which include many firms? Explain your answer.
-
Would you expect trading volume to be higher for dividend-paying stocks on the declaration date or around the cum-date/ex-date?
-
Kyrsten Haas expects an S corporation investment to generate a profit of $200,000. Her share of the S corporation is 15%. What is Kyrstens income tax if no cash is distributed? Kyrsten's marginal tax...
-
In 2000, AOL acquired Time Warner in a deal valued at $160 billion, excluding assumed debt. Time Warner is the world's largest media and entertainment company, whose major business segments include...
-
Define each of the following import policies, and describe its likely effects on the well-being of the importing country as a whole: (a) Product standards and (b) Domestic content requirements?
-
For a multi-component system, the chemical potential is equivalent to (a) Molar free energy (b) Molar concentration difference (c) Molar free energy change (d) Partial molar free energy
-
CVP exercises the Super Donut owns and operates six doughnut outlets in and around Kansas City. You are given the following corporate budget data for next year: Revenues $10,000,000 Fixed costs $...
-
Given the extensive problems with the CAPM assumptions, why is it considered the best method for computing cost of equity? Do you agree with this? What alternatives exist? What can/should a financial...
-
Exercise 2: Implement calculate_S and calculate_R Recall that the element-wise squared Euclidean distance matrix D O DE Rnxm is defined by [D D] = (x; zj) (x; Zj) - - Also, the matrices S, RE Rnxm...
-
The following rearrangement was devised and carried out to prove the stereochemistry of [1, 5] sigma tropic hydrogen shifts. Explain how the observed result confirms the predictions of...
-
When the compound having a Cyclobutene fused to a five-membered ring is heated, (IZ, 3Z)-1, 3-cvcloheptadicne is formed. When the related compound having a Cyclobutene fused to an eight-membered ring...
-
A three-phase transformer is rated \(1000 \mathrm{MVA}, 220 \mathrm{Y} / 22 \Delta \mathrm{kV}\). The Y-equivalent short-circuit impedance, considered equal to the leakage reactance, measured on the...
-
Christina Christo, the 21 year-old daughter of James Earl Paiva's former wife, testified that in 1983 she discovered a plastic bag containing white powder in one of Paiva's shoes. Christina testified...
-
According to Mitchell v. Wisconsin , did Officer Green need a warrant prior to ordering the blood draw at the hospital? Why or why not?
-
Discuss whether Caitlin Jones's case has any similarities with the Mitchell v. Wisconsin case. Are the two cases dissimilar in any way?
-
List down and explain 5 laws that explains about EXCLUSION CLAUSE. For each law, explain what has been stated and how it is being implemented. make sure to reference and footnote in Oscola.
-
Describe two strategies that would address complaints resolution processes. Identify and discuss how these also address consumer rights and responsibilities.
-
During an action potential, (a) the membrane potential becomes more negative until it hits threshold. (b) the membrane potential increases as potassium ions flow out. (c) the membrane potential...
-
Which should drive action planning more, strengths or weaknesses? That is, is it more important to build on your strengths or to reduce your weaknesses? Explain.
-
What is the difference between an immediate or cancel order and a flash order?
-
Outline efficient syntheses of each of the following alkynes from acetylene and any necessary organic or inorganic reagents: (a) 2-Heptyne (b) 3-Heptyne
-
Which of the alkynes of molecular formula C5H8 can be prepared in good yield by alkylation or dialkylation of acetylene? Explain why the preparation of the other C5H8 isomers would not be practical.
-
Give the structures of three isomeric dibromides that could be used as starting materials for the preparation of 3,3-dimethyl-1-butyne.
-
3) Two masses are connected with a thin string over a massless, frictionless pulley. If the 12.0 kg box descends 14.0 m in 8.00 seconds after being released from rest, draw two free body diagrams and...
-
For this problem, consider a model of the hydrogen atom where the "atom" is an electron confined to be in a one-dimensional box. Take the length of the box, L, to be twice the Bohr radius ao. The...
-
A floating ice block is pushed through a displacement a = (23.0 m ) - (22.0 m ), along a straight embankment by rushing water, which exerts a force = (266 N )i (186 (186 N ); on displacement? on the...

Study smarter with the SolutionInn App


