The following reactivity order has been found for the basic hydrolysis of p-substituted methyl benzoates: Y =
Question:
The following reactivity order has been found for the basic hydrolysis of p-substituted methyl benzoates: Y = NO2 > Br > H > CH3 > OCH3. How can you explain this reactivity order? Where would you expect Y = C ? N = Y = CHO, and Y = NH2 to he in the reactivity list?
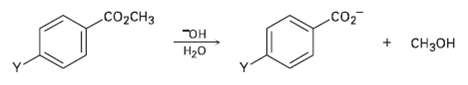
Transcribed Image Text:
co Со2CHз Dн CHзон H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
A negatively charged tetrahedral intermediate is formed when the nucleoph...View the full answer

Answered By

Tamil Elakkiya Rajendran
I'm currently involved in the research in the field of Biothermodynamics, Metabolic pathway analysis and computational Biology. I always prefer to share my knowledge whatever I have learnt through my degree whenever time permits.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following reactivity order has been found for the saponification of alkyl acetates by aqueous NaOH. Explain. CH3CO2CH3 > CH3CO2CH2CH3 > CH3CO2CH (CH3)2 > CH3CO2C (CH3)3
-
How can you explain the fact that franc-i -hromo-2-methylcyclohcxane yields the non-Zaitsev?s elimination product 3-methylcyclohexene on treatment with base? H C Br trans-1-Bromo-2-methylcyclohexane...
-
How can you explain the Enron bankruptcy scandal as an example of Mishkin's concerns about asymmetric information problems in the securities markets?
-
What is the potential difference across one wire of a 30-m extension cord made of 16-gauge copper wire carrying a current of 5.0 A?
-
What is uptalk, and why is it unprofessional? What other poor voice habits can you identify?
-
Bird B (from Table 7.1). Find the probability distribution of W = 0.05L + 0.02M for the following birds. Check that the expectation is 0.059 in both cases.
-
Implement an edge detection system in the programming language of your choice. You will need first to find a way to obtain pixel data from an image and convert this into a two-dimensional array of...
-
On July 1, Coastal Distribution Company is considering leasing a building and buying the necessary equipment to operate a public warehouse. Alternatively, the company could use the funds to invest in...
-
The following transactions were completed by Irvine Company during the current fiscal year ended December 31: Feb. 8 Received 35% of the $18,100 balance owed by DeCoy Co., a bankrupt business, and...
-
Please watch the documentary here: https://youtu.be/hkQsHHwsj4M What is a critique? Pick 2 articles throughout the semester and critique them based on the following questions. Your critique must be...
-
Outline methods for the preparation of acetophenone (phenyl methyl ketone) starting from the following: (a) Benzene (b) Bromobenzene (c) Methyl benzoate (d) Benzonitrile (e) Styrene
-
Explain the observation that attempted Fischer esterification of 2, 4, 6-tri-methylhenzoic acid with methanol and HC1 is unsuccessful. No ester is obtained, and the acid is recovered unchanged. What...
-
Predict the major alkene product of the following E1reaction: CHBr eat CH-CH
-
Joe has been having a few good days. Recently, Joe went to a flea market and purchased an old piano for $ 1 0 0 . To his surprise, Joe learned that the piano has a fair market value of $ 1 5 , 0 0 0...
-
You decide to also write an email to Andrew Suarez, who is known as the genius behind all the successful Systech products. He is only 2 8 years old and was recently appointed R&D Manager with more...
-
It is the beginning of April and Comfy Home's accountant has gone away for a month to care for a sick family member. Tenisa really wants to know how the business fared in the first quarter. She has...
-
El costo de adquisicin de los activos fijos siempre se mantiene en los libros en su balance original, cuando se disponen se elimina el balance (costo) original y la depreciacin acumulada. Datos:...
-
On January 1, 2023, your company acquires 10,000 shares of Investee Business Limited (IBL), representing 41% of the shares of IBL, for $202,000. As part of that investment, your company is entitled...
-
Why do rocks made from slowly cooling magma have large crystals? Give an example of this kind of rock.
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
Determine the geometry about each interior atom in each molecule and sketch the molecule. (Skeletal structure is indicated in parentheses.) a. CH 3 NH 2 (H 3 CNH 2 ) b. CH 3 CO 2 CH 3 (H 3 CCOOCH 3...
-
Give the principal products expected when 4-octyne or the other compounds indicated are treated with each of the following reagents: (a) H2, Pd/C catalyst (b) H2, Lindlar catalyst (c) Product of (b)...
-
In its latest catalog, Blarneystyne, Inc., a chemical company of dubious reputation specializing in alkynes, has offered some compounds for sale under the following names. Although each name...
-
Build a model of cyclodecyne. Compare its stability qualitatively to that of cyclohexyne; explain your answer.
-
Consider the general case of a block of mass MB hanging at rest and a dart of mass mo moving horizontally with speed vo. The dart hits and embeds in the block, and the center of mass of the...
-
3 kg of water, originally at 10 degrees Celsius, is mixed with 10 kg of lead. The final result at equilibrium is 16 degrees Celsius. Find the initial temperature of the lead. C = 4186 kg-C (specific...
-
Write a java program to perform to find out area of a square?

Study smarter with the SolutionInn App


