The glycolysis pathway shown in figure has a number of intermediates that contain phosphate groups. Why can
Question:
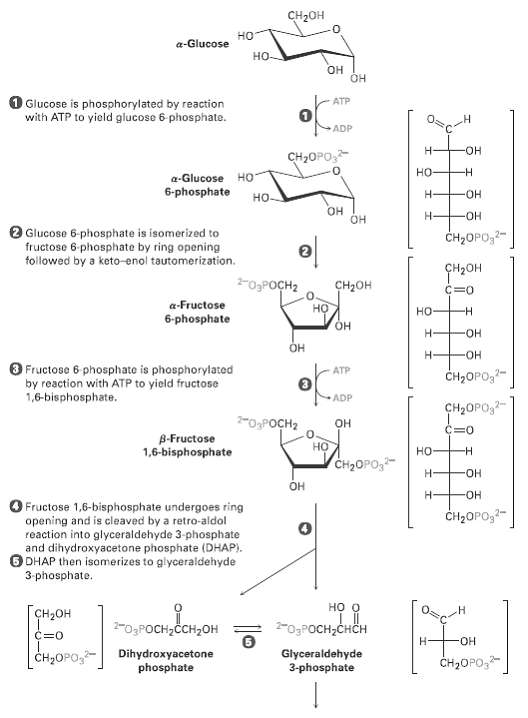
The glycolysis pathway shown in figure has a number of intermediates that contain phosphate groups. Why can 3-phosphoglyceryl phosphate and phosphoenolpyruvate transfer a phosphate group to ADP while glucose 6-phosphatecannot?
Transcribed Image Text:
CH2он но a-Glucose но- он OH O Glucose is phosphorylated by reaction with ATP to yield glucose 6 phosphate. ATP ADP н- HO- сн-OPО, но a-Glucose HO 6-phosphate H- HO- но- Он он -HO- Glucose 6-phosphate is isomerized to fructose 6 phosphate by ring opening tollowed by a keto-enol tautomerization. CH2OPO,2- ҫнрон -O3POCH2 CH2он C=0 a-Fructose но но 6-phosphate Н HO- Он ОН O Fructose 6 phosphate is phosphorylated by reaction with ATP to yield fructose 1,6-bisphosphate. ATP CH2OPO,?- ADP CH2OPO,- 2-OPOCH2 он C=0 B-Fructose 1,6-bisphosphate но но -н CH2OPO, Н HO- Он ОН O Fructose 1,6-bisphosphate undergoes ring opening and is cleaved by a retro-aldol reaction into glyceraldehyde 3-phosphate and dihydroxyacetone phosphate (DHAP). DHAP then isomerizes to glyceraldehyde CH2OPO,- 3-phosphate. но о Гсн-он -0,Росн-снсн 2-O3POCH,ČCH,OH C=0 HO- H- Dihydroxyacetone phosphate Glyceraldehyde 3-phosphate | Cн-ОPО CH2OPO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
As we saw in Section 291 formation of glucose 6phosphate from glucose and ATP i...View the full answer

Answered By

Anjali Arora
Having the experience of 16 years in providing the best solutions with a proven track record of technical contribution and appreciated for leadership in enhancing team productivity, deliverable quality, and customer satisfaction. Expertise in providing the solution in Computer Science, Management, Accounting, English, Statistics, and Maths.
Also, do website designing and Programming.
Having 7 yrs of Project Management experience.
100% satisfactory answers.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The oven shown in Figure has a heating element with appreciable capacitance C1. The other capacitance is that of the oven air C. The corresponding temperatures are T1 and T2, and the outside...
-
The biosynthetic pathway shown in Figure 26.10 was developed with the aid of isotopic labeling experiments. Which carbon atoms of cholesterol would you expect to be labeled when acetate enriched with...
-
The biosynthetic pathway shown in Figure 26.10 was developed with the aid of isotopic labeling experiments. Which carbon atoms of cholesterol would you expect to be labeled when acetate enriched with...
-
This chapter describes the mechanisms in place to regulate accounting and financial reporting in five countries. Required: Compare and contrast these mechanisms in the United Kingdom and China.
-
What are the differences between GAAP based and Pro Forma financial statements?
-
Apply the full White test for heteroskedasticity [see equation (8.19)] to equation (8.18). Using the chi-square form of the statistic, obtain the p-value. What do you conclude?
-
The \(T d S\) equation \(T d S=C_{P} d T-T\left(\frac{\partial V}{\partial T} ight)_{P} d P\) is valid where entropy is a function of (a) \(T\) and \(P\) (b) \(P\) and \(V\) (c) \(T\) and \(V\) (d)...
-
The following describes the expenditure cycle manual procedures for a hypothetical company. The inventory control clerk examines the inventory records for items that must be replenished and prepares...
-
What are the psychological and emotional dimensions of organizational commitment, and how can organizations address these dimensions to foster deeper employee loyalty?
-
Your marketing research firm is preparing the final written report on a research project commissioned by a major manufacturer of lawn mowers. One objective of the project was to investigate seasonal...
-
What enzyme cofactor is associated with each of the following kinds of reactions? (a) Transamination (b) Carboxylation of a ketone (c) Carboxylation of an -keto acid
-
In the pentose phosphate pathway for degrading sugars, ribulose 5-phosphate is converted to ribose 5-phosphate. Propose a mechanism for theisomerization. CH2 - HO- C=0 HO- - - HO- -OH H. -HO- -...
-
The file contains the state cigarette tax ($) for each state as of December 31, 2009. a. Construct an ordered array. b. Plot a percentage histogram. c. What conclusions can you reach about the...
-
The infamous chicken is dashing toward home plate with a speed of 5.10 m/s when he decides to hit the dirt. The chicken slides for 1.25 s, just reaching the plate as he stops. What is the magnitude...
-
An electron enters a region of space containing a uniform 2.77 10-5 T magnetic field. Its speed is 135 m/s and it enters perpendicularly to the field. Under these conditions, the electron undergoes...
-
An unbanked (flat) curve of radius 150 m is rated for a maximum speed of 29.0 m/s. At what maximum speed, in m/s, should a flat curve with radius of 63.0 m be rated? 63 Consider the relationship...
-
The magnetic coils of a tokamak fusion reactor are in the shape of a toroid having an inner radius of 0.700 m and an outer radius of 1.30 m. The toroid has 840 turns of large-diameter wire, each of...
-
Two masses are arranged as in the diagram with an angle of 30. The mass of m is 1.5x times m2. What is the minimum coefficient of friction so that m does not go towards the ground (or ml up the ramp)...
-
Can two species have the exact same niche in a community? Why or why not?
-
Write out the formula for the total costs of carrying and ordering inventory, and then use the formula to derive the EOQ model. Andria Mullins, financial manager of Webster Electronics, has been...
-
Redo Problem 14.8 using Aspen Plus. Problem 14.8 Methane is to be burned in air. Determine the adiabatic flame temperature as a function of the methane-to-air ratio at a pressure of 1 bar.
-
The two dimers of 2-methylpropene shown in the equation can be converted to 2,2,4-trimethylpentane (known by its common name isooctane) for use as a gasoline additive. Can you suggest a method for...
-
Write the structure of the major organic product formed in the reaction of 1-pentene with each of the following: (a) Hydrogen chloride (b) Hydrogen bromide (c) Hydrogen bromide in the presence of...
-
Repeat Problem 6.22 for 2-methyl-2-butene. In Problem 6.22 Write the structure of the major organic product formed in the reaction of 1-pentene with each of the following: (a) Hydrogen chloride (b)...
-
Golden Gate Mining Co. (GGMC) currently has a gold mine operating in Canada and is looking to purchase another gold mine in Peru, operating as Inca Gold Inc. (ICI). You currently work for the CFO at...
-
Cokolo Inc. has entered into the following two derivatives contracts: Purchased put options on 1,000 Trenton Co. shares that have an exercise price of $52 per share and expire in 60 days Negotiated a...
-
As a financial manager (consultant), undertake an investigation on your allocated company to evaluate its strategic financial position. Prepare a business report for the board of directors of the...

Study smarter with the SolutionInn App


