The hydrogen's of the hydrocarbons can b randomly replaced by chlorine by reaction of the hydrocarbon with
Question:
The hydrogen's of the hydrocarbons can b randomly replaced by chlorine by reaction of the hydrocarbon with Cl2 in the presence of light. An unknown compound, A, with the formula C4H10, produces two isomeric monochlroides, B and C, with the formula C4H9Cl, when submitted to these reaction conditions. Monochlroides B produces three isomeric dichlorides, D, E, and F, with the formula C4H8Cl2, when submitted to these reactions conditions. Monochlroides C gives a single dichloride, D, when submitted to these reaction conditions. Show the structures of A, B, C, D, E, and F. Are there any ambiguities in these structure assignments?
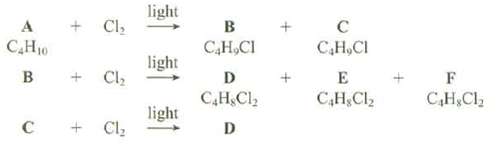
light + Cl, B С Н.СI САНю С.Н, СI light B Cl, E D С.Н.С, С,Н, С, С,Н, С. light Cl, D +
Step by Step Answer:
Based on these experiments it cannot be determined wh...View the full answer



Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Organic Chemistry questions
-
(a) An unknown compound A has the formula C 7 H 12 O 2 and infrared spectrum A (p. 917). To which class does this compound belong? (b) Use the other spectra (NMR-B, p. 917, and F, p. 919; IR-D, E,...
-
An unknown compound A of molecular formula of C10H180 reacts with H2S04 and heat to form two compounds (B and C) of molecular formula C10H16. B and C both react with H2 over Pd/C to form decalin....
-
An unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is...
-
A mixture of 1 kmol carbon dioxide, 2 kmol carbon monoxide, and 2 kmol oxygen, at 25C, 150 kPa, is heated in a constant pressure steady state process to 3000 K. Assuming that only these same...
-
Arlington Corporation's financial statements (dollars and shares are in millions) are provided here Income Statement for Year Ending December 31, 2016...
-
Explain the concept of non monetary rewards.
-
Incubators are organizations that provide startups with shared operating space. They are sponsored by universities, city or county governments, or nonprofit organizations. Incubators also provide...
-
Draw an entity relationship diagram (ERD) for the following situations: 1. Whenever new patients are seen for the first time, they complete a patient information form that asks their name, address,...
-
(a) (b) (c) Small business owners are discovering that social media marketing is quickly becoming an important method for driving business growth. While the idea of using "free tools" to drive...
-
Who should get the drones initially How can you justify your decision ethically What restrictions for use should these people be given, and how do you think employees, both those who get drones and...
-
To find a base that is strong enough to deprotonate benzoic acid but not p-methyl phenol. Then explain how this base might be used to separate these two compounds in the laboratory.
-
Provide structures for these naturally occurring compounds: (a) 2, 6-Diaminohexanoic acid (lysine, an amino acid) (b) Hex-2-en-1-yl acetate (sex attractant of Indian water bug) (c) (Z)-7-Dogecen-1-yl...
-
How is the demand for female labor changing in the taxi industry? Uber, along with Careem, its Middle East competitor, is hiring women drivers in Saudi Arabia after the Kingdom announced plans to...
-
How does the unfamiliar trade (forex) market influence worldwide monetary administration for a situation study, and what are the key factors that impact trade rates and money risk in worldwide...
-
Solve the following linear program using the graphical method (graph sheet on next page). Identify the decision variables. Compute the value of the decision variables and the objective function at...
-
How in all actuality does exchange highlight in worldwide monetary administration contextual analyses, and which job does it play in exploiting cost differences in worldwide business sectors?
-
1. The scores of the students in the final examination are normally distributed with a mean of 72 and a standard deviation of 8. a. Find the percentage of students who scored less than 60. b. Find...
-
1. What method are you using to track your daily spending? If you using an app, what app are you using? 2. Is your chosen method "working" for you? In other words, are you tracking your spending each...
-
All of the following are benefits of Ijarah transactions except: a. Ijarah allows companies to completely finance their longterm assets. b. Ijarah can effectively reduce the risk of obsolescence. c....
-
The Alert Company is a closely held investment-services group that has been very successful over the past five years, consistently providing most members of the top management group with 50% bonuses....
-
Suppose the government passes an immigration reform law that is successful in preventing most illegal immigrants from getting employment in the United States. Which American groups would benefit and...
-
The DNA of sea urchins contains about 32% A. What percentages of the other three bases would you expect in sea urchin DNA? Explain.
-
The codon UAA stops protein synthesis. Why does the sequence UAA in the following stretch of mRNA not cause any problems? -GCA-UUC-GAG-GUA-ACG-CCC-
-
Which of the following base sequences would most likely he recognized by a restriction (endonuclease? Explain. (a) GAATTC (b) GATTACA (c) CTCGAG
-
Complete the table by: i. Defining each type of taxable transaction data provided below. ii. Listing one unique source of information for each type of taxable transaction data listed below. iii....
-
You are a partner in a CPA firm. You have a client that is being audited by the IRS. The IRS is using an indirect method of proof of his income. Your client is an attorney but not a tax attorney and...
-
If Olivia is single and in the 24 percent tax bracket, and she sold stock for $12,000 that she purchased for $10,000, 9 months earlier. What should her tax expense be in 2018?

Study smarter with the SolutionInn App


