The integrated 1H NMR spectrum of a compound of formula C4H10O is shown in figure. Propose astructure.
Question:
The integrated 1H NMR spectrum of a compound of formula C4H10O is shown in figure. Propose astructure.
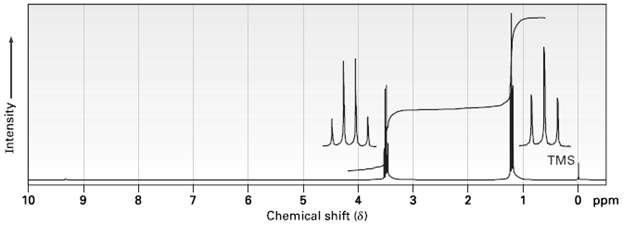
Transcribed Image Text:
TMS 10 8. 6. Chemical shift (8) 3 O ppm Intensity 6.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
The molecular formula C4H10O indicates that the compound has no ...View the full answer

Answered By

CHARLES AMBILA
I am an experienced tutor with more than 7 years of experience. I have helped thousands of students pursue their academic goals. My primary objective as a tutor is to ensure that students have easy time handling their academic tasks.
5.00+
109+ Reviews
323+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H NMR spectrum of a compound (C10H13BrO) is shown in Figure 16.10. The compound gives benzyl bromide, along with a second compound C3H6Br2, when heated with HBr. What is the first compound?
-
The 1H NMR spectrum of a compound (C10H13BrO) is shown in Figure 16.10. The compound gives benzyl bromide, along with a second compound C3H6Br2, when heated with HBr. What is the first compound?
-
The proton NMR spectrum of a compound of formula C 10 H 12 O follows. This compound reacts with an acidic solution of 2,4-dinitrophenylhydrazine to give a crystalline derivative, but it gives a...
-
From the information below, 1. List of the working capital accounts. 2. Calculate the net working capital. Buildings $100,000 Cash ... 5,000 Trade receivables ... 25,000 Trade and other payables .....
-
The large turbine generator industry is a duopoly. The two firms, GE and Westinghouse, compete through Cournot quantity setting competition. The demand curve for the industry is P=100-Q, where P is...
-
For a given substance, the liquid crystalline phase tends to be more viscous than the liquid phase. Why?
-
Define the modes of entrepreneurial finance available to entrepreneurs.
-
Preparing a bank reconciliation Harrison Photography's checkbook lists the following: Harrison's November bank statement shows the following: Requirements 1. Prepare Harrison Photography's bank...
-
How does remote work or virtual collaboration affect employees' sense of connection and commitment to an organization ?
-
Mountain View Hospital has purchased new lab equipment for $134,650. The equipment is expected to last for three years and to provide cash inflows as follows: Year 1. . . . . . . . $45,000 Year 2. ....
-
Draw structures for compounds that meet the following descriptions: (a) C2H6O; one singlet (b) C3H7Cl; one doublet and one septet (c) C4H8Cl2O; two triplets (d) C4H8O2, one singlet, one triplet, and...
-
3-Bromo-1-phenyl-1-propene Shows a complex NMR spectrum in which the vinylic proton at C2 is coupled with both the Cl vinylic proton (J = 16 Hz) and the C3 rnethylene protons (J = 8Hz). Draw a tree...
-
In Problems 31 through 42, verify that the given differential equation is exact; then solve it. 2x y 3y x4 dx + + -15 dy = 0
-
4. Assume the BSM assumptions hold. A non-dividend-paying stock has a spot price of 10, an expected return of 5% per year and a return volatility of 20% per year. The continuously- compounded...
-
Thames Health System has a target capital structure of 30 percent debt and 70 percent equity. The system's CFO has obtained the following information: (1) the before-tax yield to maturity on the...
-
York Health has $800,000 of debt outstanding, and it pays an interest rate of 10 percent annually on its bank loan. York's total revenues are $3,200,000; its average tax rate is 40 percent, and its...
-
Assume perfect capital markets. Roche Technology's stock has a beta of 1.2 and has an expected return of 12.5%. It is a zero-debt firm. Suppose it issues new risk- free debt with a cost of debt of 5%...
-
Krugman Technology is currently a zero-debt firm with a stock price of $7.50 per share and 20 million shares. Although investors currently expect Krugman to remain a zero-debt firm, Krugman plans to...
-
Which is the best representation in Figure P10.39 of the trajectory of a cantaloupe thrown horizontally off a bridge? What is wrong with the other paths? Data from Figure P10.39 W
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
An ethylene glycol solution contains 21.2 g of ethylene glycol (C 2 H6O 2 ) in 85.4 mL of water. Determine the freezing point and boiling point of the solution.
-
For each spectrum, interpret all the significant stretching frequencies above 1580 cm-1. wavelength (um) 2.5 100 4 4.5 9 10 12 13 14 15 16 60 N 40 T 1642 4000 3500 3000 2500 2000 1800 1600 1400 1200...
-
Point out which of these four mass spectra indicate the presence of sulfur, chlorine, bromine, iodine, or nitrogen. Suggest a molecular formula for each? 100 156 158 80 40 20 0 10 2030 40 50 60 70 80...
-
Show the fragmentation that accounts for the cation at m/z 57 in the mass spectrum of 2-methylpentane. Explain why this ion is less abundant than those at m/z 71 and 43?
-
1. Write a reflection on advantages and disadvantages of a major decision you took a. A self-evaluation the advantages and disadvantages (on a scale from 1-10). b. A justification for your...
-
BBF315/05 Risk Management May 2023 Assignment 2 Question 1 (a) Risk management is sometimes described as the process of identifying and evaluating the trade-off between risk and expected return, and...
-
A 1.0000x10 -11 m wavelength x-ray photon strikes an atom in an example of Compton scattering. The scattered photon has a wavelength of 1.0301x10 -11 m at an angle of 29 o from the initial direction....

Study smarter with the SolutionInn App


