The Knoevenagel reaction is a carbonyl condensation reaction of an ester with an aldehyde or ketone to
Question:
The Knoevenagel reaction is a carbonyl condensation reaction of an ester with an aldehyde or ketone to yield an ?, ??un-saturated product. Show the mechanism of the Knoevenagel reaction of diethyl malonate with benzaldehyde.
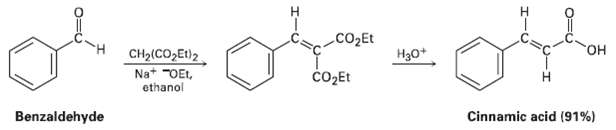
Transcribed Image Text:
cO2Et H30+ HO. CH2(CO,Et)2 čO2Et Na+ "OEt, ethanol Cinnamic acid (91%) Benzaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Formation of the enolate of diethyl malonate is the ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
An ester is a compound formed by a condensation reaction between a carboxylic acid and an alcohol. Read the discussion of esters in Section 24.4 and then give an example of a reaction forming an...
-
The reaction of an aldehyde or ketone with a Grignard reagent is a nucleophilic addition to the carbon-oxygen double bond. (a) What is the nucleophile? (b) The magnesium portion of the Grignard...
-
Diethyl malonate is prepared commercially by hydrolysis and esterification of ethyl cyanoacetate. The preparation of ethyl cyanoacetate proceeds via ethyl chloroacetate and begins with acetic acid....
-
We look at the accumulated area beneath this curve, as in the definite integral as follows F(x) = f(t) dt -2 { F(x) = int_(-2)^x f(t) dt a) Use ordinary area formulas to compute each of the...
-
Give examples of actions that are substitutes in production. Give examples of actions that are compliments in production.
-
Write equations that show the processes that describe the first, second, and third ionization energies of an aluminum atom. Which process would require the least amount of energy?
-
What is the purpose of the IT strategic planning process?
-
Salmons Stores operates a national chain of women's apparel stores. Five thousand copies of an expensive four-color sales catalog have been printed, and each catalog includes a coupon that provides a...
-
A. Describe what are: External Assessment' and 'Internal Assessment' B. What does 'external assessment' focus on? What does 'internal assessment' focus on?
-
1. Estimate the final construction budget. 2. What should they do about the work slowdown? 3. Create a Gantt chart with the revisions to the duration of the interiors step. When will the project be...
-
The amino acid leucine is biosynthesized from a-ketoisovalerate by the following sequence of steps. Show the mechanism ofeach. Acetyl CoA COASH NAD+ NADH/H* CO2 "C 2-Isopropylmalate co2 ...
-
The Darzens reaction involves a two-step, base-catalyzed condensation of ethyl chloroacetate with a ketone to yield an epoxy ester. The first step is a carbonyl condensation reaction, and the second...
-
$15,000 was invested, from which can be withdrawn a geometric gradient series of annual payments decreasing at the rate of 10% per year. The first payment received was $3,000 and it occurred after...
-
The MC company presents the following information for 2022. There is an excess in tax depreciation over book depreciation of $40,000. This difference will be reversed in equal parts in 2023 to 2026....
-
What strategies does an RTOS employ to address priority inversion? Can you describe a scenario where priority inversion could cause critical issues and how the RTOS mitigates it ?
-
Determine the Prior Year and Current Year trend percents for net sales using the Prior Year as the base year. Note: Enter the answers in thousands of dollars.
-
Discuss the gender wage gap. What are current estimates in the United States and what are some potential reasons for the wage gap?
-
The Super Shine Furniture Polish Company has provided you the following information for the year 2 0 2 3 : Cash $ 1 4 , 4 0 0 Accounts receivable 2 3 , 1 0 0 Accumulated depreciation - equipment 2 0...
-
Estimate the rotational inertia of a \(70-\mathrm{kg}\) athlete about a horizontal axis passing through her waist from left to right.
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
Use the BornHaber cycle and data from Appendix IIB, Chapter 9, and this chapter to calculate the lattice energy of KCl. (H sub for potassium is 89.0 kJ/mol.) Substance Aluminum Al(s) Al(g) Al+(aq)...
-
Give the expected major product for each reaction, including stereochemistry where applicable. (a) but-1-ene + H2 / Pt (b) cis-but-2-ene + H2 / Ni (c) (d) + H/Pt + excess H,/Pt
-
One of the principal components of lemon grass oil is limoneneC10H16 When limonene is treated with excess hydrogen and a platinum catalyst, the product is an alkane of formula C10H20. What can you...
-
The chiral BINAP ligand shown in Figure 8-7 contains no asymmetric carbon atoms. Explain how this ligand is chiral.
-
Suppose the amounts presented here are basic financial information (in millions) from the 2025 annual reports of Nike an Nike adidas Sales revenue $18,455.5 10,493.0 Allowance for doubtful accounts,...
-
You can afford a $250 per month car payment. You\'ve found a 5year loan at 5% interest. How big of a loan can you afford?
-
When Karina was 6 years old, her parents started depositing $1,200 every three months into an account in Bank Mitch that pays interest at the rate of 1.75%/year compounded quarterly until she gets...

Study smarter with the SolutionInn App


