The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound,
Question:
The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound, what is the structure of the ion responsible for the peak at m/z 43 in eachspectrum?
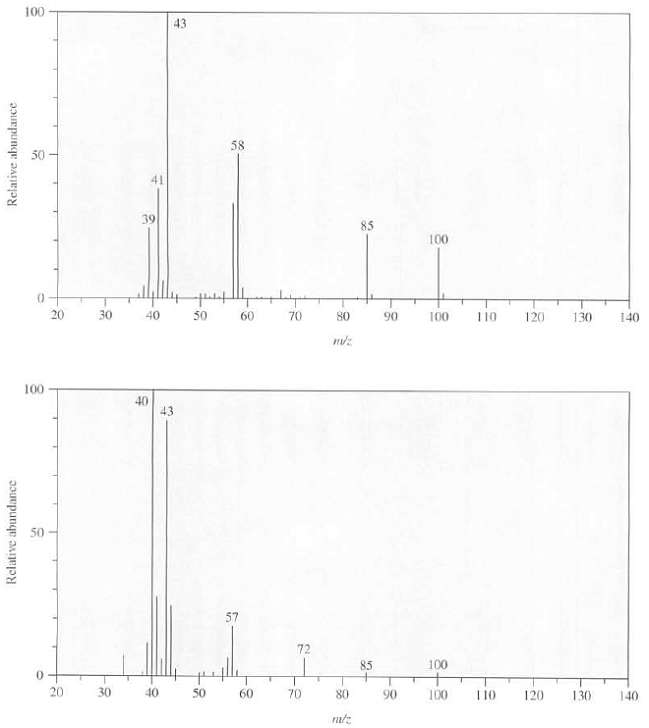
Transcribed Image Text:
100 43 58 50 41 39 85 100 20 30 40 50 70 60 80 90 100 110 120 130 140 100 40 43 57 72 100 85 20 30 40 50 60 70 80 90 100 T10 120 130 140 Relative abundance Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
Both the ketones produce fragment ions at mz 43 and 85 due t...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mass spectra of 1-methoxybutane, 2-methoxybutane, and 2-methoxy-2-methylpropane are shown in Figure 13.7. Match the compounds with the spectra. 100 73 80 S 60 57 20 0 10 20 30 40 50 60 70 80 90...
-
The mass spectra of two very stable cycloalkanes both show a molecular ion peak at m/z = 98. One spectrum shows a base peak at m/z = 69, the other shows a base peak at m/z = 83. Identify the...
-
The mass spectra of acid derivatives follow the principles shown in Chapter 18 for other carbonyl compounds and for alkoxy groups. Both McLafferty rearrangements and alpha-cleavages are common. The...
-
Holly needs $21,800 worth of new equipment for his shop. He can borrow this money at a discount rate of 11% for a year. Find the amount of the loan Holly should ask for so that the proceeds are...
-
IBM has a bond issue outstanding with 14 years to maturity. When originally issued the bond had a par value of $1,000, a stated coupon rate of 12% and 15 years to maturity. Currently, similar risk...
-
One month after the restructuring, Brooks is informed by the EEOC that, thus far, four complaints have been file against Brooks for discrimination in the process. What data should you examine to...
-
This chapter has identified a range of stakeholders that managers should consider when determining sustainability performance and reporting. Determine how managers should engage with each of these...
-
In 1990, the city of Worcester, Massachusetts, adopted an ordinance that required rooming houses to be equipped with automatic sprinkler systems no later than September 25, 1995. In Worcester, James...
-
4. 5. In which of following conditions a real gas would behave ideally? (a) Low pressure and low temperature ha (b) At value of temperature equal to its Boyle's temperature (c) Between its critical...
-
Assume that the shortest distance between nodes a, b, c, and d no node y and the costs from node x to nods a, b, c, and d are given below: Day = 5, Dby = 6, Dcy = 4, Ddy = 3 cxa = 2, cxb = 1, cxc =...
-
Suggest a structure for the compound whose mass spectrum is asfollows: 100 107 81 93 188 70 80 110 120 90 100 130 150 160 170 140 180 190 200 Relative abundance
-
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm ?1 in its IR spectrum and B has a peak at 1715cm ?1 , the mass spectra of A and B are as follows, show the structures of...
-
If this is a product you have actually purchased, explain the reasoning behind your current satisfaction or dissatisfaction with the product. If not, develop several reasons you might be satisfied or...
-
Describe a situation in which you experienced two decision-making pitfalls. What techniques could be applied in each situation for more effective decision-making? Which decision-making techniques or...
-
A cement block of mass 20.0 kg is at rest on an inclined plane that makes an angle q=30.0 with the horizontal. What is the normal force acting on the block? = 9.81 m/s2 [down] 196 N [270] b. 170 N...
-
The gas phase decomposition of sulfuryl chloride at 600 K SOCl (9) SO(9)+ Cl2(9) is first order in SO2 Cl with a rate constant of 2.80 10-3 min. If the initial concentration of SO2Cl2 is 0.000890 M,...
-
Conduct a coaching session Your coaching sessions need to include the following elements and is to be run in the following order: TASK 1 - Explanation: You are to explain what you will be...
-
In the figure, all the charges are point charges and the charge in the middle is Q=-3.1 nC. For what charge q: will charge qe be in static equilibrium? 91 2 10 cm 10 cm
-
In 1989, New Zealand rewrote the charter of its central bank to make low inflation its only goal. Why would New Zealand want to do this?
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
In late November and early December 2006, there was speculation in the financial markets as to what the Federal Reserve planned to do with interest rates. After raising the federal funds interest...
-
Trichloromethane (CHCl3, also called chloroform) has a larger dipole moment than CFCl3. Use three-dimensional structures and bond moments to explain this fact.
-
Indicate the direction of the important bond moments in each of the following compounds (neglect C-H bonds). You should also give the direction of the net dipole moment for the molecule. If there is...
-
Write structural formulas for all of the alkenes with (a) The formula C2H2Br2 and (b) The formula C2Br2Cl2. In each instance designate compounds that are cis-trans isomers of each other. Predict the...
-
How does Ozy media differentiate itself from competitor? or how does it build brand identity?
-
Moe Glee's business acquired a group of assets two years ago to help with expansion plans, but Moe would now like to review the use of some of those assets. Moe's close friend Ana Maui Harry's has...
-
Analyze the centrality of Off Whites' significant collaborations with other luxury and sportswear brands for accruing valuable brand equity.?

Study smarter with the SolutionInn App


