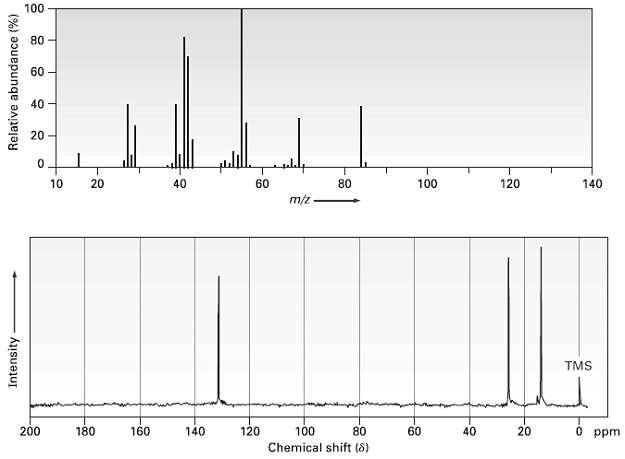
The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this
Question:
The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this hydrocarbon, and explain the spectraldata.
Transcribed Image Text:
100 80 60 40 20 aly 120 10 40 60 80 100 140 m/z TMS 200 180 160 120 100 80 60 20 0 ppm 140 40 Chemical shift (8) Intensity Relative abundance (%) 20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
The peak in the mass spectrum at mz 84 is probably the molecular ion of the unknown compound and co...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The proton and 13C NMR spectra of a compound of formula C4H11N are shown here. Determine the structure of this amine, and give peak assignments for all of the protons in the structure. 200 180 160...
-
An unknown, foul-smelling hydrocarbon gives the mass spectrum and infrared spectrum shown. (a) Use the mass spectrum to propose a molecular formula. How many elements of unsaturation are there? (b)...
-
The mass spectrum and infrared spectrum of an unknown compound are shown in Figures 13.27 and 13.28, respectively. Identify the compound. Figure 13.27 The mass spectrum for Problem 28. 100 E 80 3 60...
-
The Holtz Corporation acquired 80 percent of the 100,000 outstanding voting shares of Devine, Inc., for $7.20 per share on January 1, 2014. The remaining 20 percent of Devines shares also traded...
-
It is estimated that a firm contemplating entering the breakfast cereal market would need to invest $100 million to build a minimum efficient scale production plant (or about $10 million annually on...
-
A uniform volume charge density of 80 C/m 3 is present throughout the region 8 mm < r < 10 mm. Let = 0 for 0 < r < 8 mm. (a) Find the total charge inside the spherical surface r = 10 mm. (b) Find...
-
In addition to the size variables, we also have information on several binary variables. The variable URBAN is used to indicate the facility's location. It is one if the facility is located in an...
-
On December 31, 2017, Ball Company leased a machine from Cook for a 10-year period, expiring December 30, 2027. Annual payments of $100,000 are due on December 31. The first payment was made on...
-
The table shows the median income level for various age groups, based on the most recent U.S. Census data available. T O Median Income Age group (in years) 21-24 25-34 35-44 $38,145.00 45-54 55-64...
-
Liquid octane (C8H18) enters a steady-flow combustion chamber at 25oC and 1 atm at a rate of 0.25 kg/min. It is burned with 50 percent excess air that also enters at 25oC and 1 atm. After combustion,...
-
Propose structures for the three compounds whose 1H NMR spectra are shown. (a) C 5 H 10 O (b) C 7 H 7 Br (c) C 8 H 9 Br TMS O ppm 10 Chemical shift (8) TMS O ppm 10 9. 8. 3 2 Chemical shift (8) TMS O...
-
Compound A, a hydrocarbon with M + = 96 in its mass spectrum, has the 13 C spectral data that follow. On reaction with BH 3 followed by treatment with basic H 2 O 2 , A is converted into B, whose 13...
-
Why do we prefer to use semiconductor crystals that contain as small a number of dislocations as possible?
-
There were 417 cell phones sold at an electronics store in January. Since then, cell phone sales at this store have increased at a rate of 3.75% per month. At this rate of growth, which function can...
-
Business proposal to Sears board of directors to revitalize the organization through some well-thought-out, innovative strategies. Your proposal should include the following: An analysis of the...
-
Why is it important for Delta Lloyd Group to comply with industry regulations? What is FEMA and what does it do? What are the main challenges that FEMA faces? How did FEMA improve its inefficient...
-
How does conflict theory illuminate the role of economic inequality, class exploitation, and capitalist modes of production in perpetuating social conflict and stratification?
-
How does conflict theory intersect with other sociological theories, such as feminist theory, critical race theory, and postcolonial theory, in offering insights into systems of oppression and...
-
Acidity and alkalinity of soil are typically measured on the \(\mathrm{pH}\) scale, which has a range from \(0-14\). A pH of 7 is considered neutral, a \(\mathrm{pH}\) of \(0-7\) is acidic, and a...
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
Potassium nitrate has a lattice energy of -163.8 kcal/mol and a heat of hydration of -155.5 kcal/mol. How much potassium nitrate has to dissolve in water to absorb 1.00 * 10 2 kJ of heat?
-
Show how Diels-Alder reactions might be used to synthesize the following compounds. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH 3COOCH CH3 CN CN CI CI Cl Cl Cl CI CI C chlordane CI CI Cl Cl CI C aldrin CN...
-
Furan and maleimide undergo a Diels-Alder reaction at 25 °C to give the endo isomer of the product. When the reaction takes place at 90 °C, however, the major product is the exo isomer....
-
(a) Sketch the pi molecular orbitals of hexa-1, 3, 5-triene (Figure 15-25). (b) Show the electronic configuration of the ground state of hexa-1, 3, 5-triene. (c) Show what product would result from...
-
What are some features of Cross Laminated Timber?
-
The 201 1 price implied by PPP for the Billy bookcase in the United Kingdom was 137.49, but the actual price at that time was $29.90. What are possible reasons why the price was relatively low?
-
Within the intricate tapestry of advanced organizational ecosystems, characterized by their emphasis on continuous learning and adaptive capabilities, what strategies can organizational leaders...

Study smarter with the SolutionInn App


