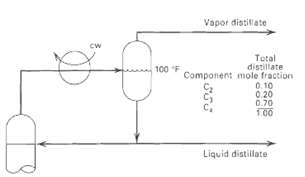
The overhead system for a distillation column is shown in Figure. The composition of the total distillates
Question:
The overhead system for a distillation column is shown in Figure. The composition of the total distillates is indicated, with 10 mol% of it being taken as vapor. Determine the pressure in the reflux drum, if the temperature is 100?F. Use the following K-values by assuming that K is inversely proportional topressure.
Transcribed Image Text:
Vapor distitlate cw Total distillate Component mole fraction 0. 10 0.20 0.70 100 100 F Liquid distillate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
As shown in Exercise 424 the compositions of net equi...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
A temperature control system for a distillation column is shown in figure. The temperature T of a tray near the top of the column is controlled by adjusting the reflux flow rate R. Draw a block...
-
A system with unity feedback is shown in Figure E5.11. Determine the steady-state error for a step and a ramp input when Figure E5.11 Unity feedback system. 5(s + 8) s(s 1)(s 4)(s 10) G(s) Gs) Yts)
-
A system with a controller is shown in Figure AP5.5. The zero of the controller may be varied. Let α = 0, 10, 100. (a) Determine the steady-state error for a step input r(t) for...
-
Name the five levels of the Capability Maturity Model.
-
At January 1, 2012, Underwood Imports Inc. reported the following on its statement of financial position: Accounts receivable...........................................$1,990,000 Allowance for...
-
What value is contained in the floating point variable depth after the following statements are executed?
-
Consider a family of call options on a non-dividend-paying stock, each option being identical except for its strike price. The value of the call with strike price \(K\) is denoted by \(C(K)\). Prove...
-
Assume that Samsung manufactures and sells 60,000 units of a product at $11,000 per unit in domestic markets. It costs $6,000 per unit to manufacture ($4,000 variable cost per unit, $2,000 fixed cost...
-
Dana's Ribbon World makes award rosettes. Following is information about the company: Variable cost per rosette Sales price per rosette Total fixed costs per month Required: $ 2.00 5.00 1500.00 1....
-
Bentley Hamilton throws the business section of The New York Times onto the conference room table and watches as his associates jolt upright in their overstuffed chairs. Mr. Hamilton wants to make a...
-
The following stream is at 200 psia and 200?F. Determine whether it is a subcooled liquid or a superheated vapor, or whether it is partially vaporized, without making a flashcalculation. Component...
-
Determine the phase condition of a stream having the following composition at 7.2oC and 2,620 kPa.Perform the calculations with a computer simulation program using at least three different options...
-
In a mixture of the two gases, the partial pressures of CH 4 (g) and O 2 (g) are 0.175 atm and 0.250 atm, respectively. a. What is the mole fraction of each gas in the mixture? b. If the mixture...
-
What is the best type of message to reach todays consumer?
-
What are the four different market segments? Why is it important to define and understand each?
-
Describe how factors shaping your identity contribute to health outcomes.
-
What are the benefits of having a two-way dialogue between buyer and seller?
-
Explain why theory is used in health communication planning, citing a specific example from the chapter.
-
Libby Petroleum incurred and paid the following costs during 2019: REQUIRED: Record the above transactions. Lease A Unproved Lease B Unproved Lease C Proved Purchased Lease D Proved Purchased...
-
Find the cross product a x b and verify that it is orthogonal to both a and b. a = (t, 1, 1/t), b = (t 2 , t 2 , 1)
-
Indicate two situations in which an alpha measure based on the global three-factor model, which includes the world market portfolio, global size, and value factors, may fail to correctly estimate the...
-
Solve the nine simultaneous linear equations below, which have a block-tridiagonal-matrix structure, by the Thomas algorithm: x2 + 2x3 +2x4+x6 = 7 X + x3 + x4 + 3x5 = 6 x + x + x3 + x5 + x6 = 6 X4 +...
-
One thousand kmol/h of a saturated-liquid mixture of 60 mol% methanol, 20 mol% ethanol, and 20 mol% n-propanol is fed to the middle stage of a distillation column having three equilibrium stages, a...
-
Does the SR method use tridiagonal-matrix equations? How does the SR method differ from the BP method? For what types of problems is the SR method preferred over the BP method? What are the tear...
-
How do the resource descriptions (typing) used in the Incident Command System lead to a more effective response?
-
A company incurs $4172000 of overhead each year across three departments: Ordering and Receiving, Mixing, and Testing. The company prepares 2000 purchase orders, works 50000 mixing hours, and...
-
Hemming Company reported the following current-year purchases and sales for its only product. Date January 1 January 10 Activities Beginning inventory March 14 March 15 July 30 Sales Purchase Sales...

Study smarter with the SolutionInn App


