The pKa for phenol is 10, the pKa for ethanol is 16, and the pKa for carbonic
Question:
The pKa for phenol is 10, the pKa for ethanol is 16, and the pKa for carbonic acid (H2CO3) is 6.35. Complete these equations and predict whether the reactants or the products are favored atequilibrium.
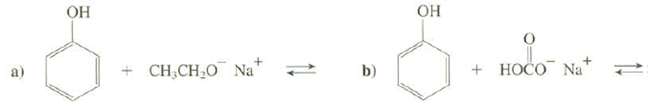
Transcribed Image Text:
OH OH + CH,CH O Na* + HOČO Na b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
In acidbase reactions the equilibrium favors the formation of the weake...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions. (a) Phenol + acetic anhydride (b) Phenol + acetic formic anhydride (c) Aniline + phthalic anhydride (d) Anisole + succinic anhydride and aluminum...
-
Predict the products of the following acid-base reactions. Using pKa values, indicate which side of each equilibrium reaction is favored, and label the species representing the stronger acid and...
-
The acid HA has pKa = 4.00. Use Equations 9-17 and 9-18 to find the fraction in the form HA and the fraction in the form A- at pH = 5.00. Does your answer agree with what you expect for the quotient...
-
How does feedback value relate to predictive ability and accountability?
-
Matthew sold 200 shares of Xerox stock on December 23, 2012. The stock had a basis of $4,000 and was sold for $2,100. When Matthew funds his IRA on January 3, 2013, he causes the IRA to purchase 400...
-
A 0.61-m copper rod with a mass of 0.043 kg carries a current of 15 A in the positive x direction. What are the magnitude and direction of the minimum magnetic field needed to levitate the rod?
-
Consider the situation of Example 12.10. Rather than shorting a futures contract, the U.S. firm could borrow \(500 /\left(1+r_{G} ight)\) euros (where \(r_{G}\) is the 90-day interest rate in...
-
Nick Company had the following transactions during April. 1. Sold merchandise on account. 2. Purchased merchandise on account. 3. Collected cash from a sale to Athletic Company. 4. Recorded accrued...
-
Required Information [The following Information applies to the questions displayed below.] Warnerwoods Company uses a perpetual Inventory system. It entered into the following purchases and sales...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
Show the resonance structures for the conjugate base of phenol.
-
Provide names for thesecompounds: a) CH;CH,CH,CH,CH,CH b) CH f) CH,CCH,CCH, d) CI g) h)
-
In Problems 17 and 18, find the value of each determinant. -2 5 3 7
-
CFO of AAA company estimates the following spot interest rates on Treasure Securities: r 1 = 4 % , r 2 = 5 % , r 3 = 5 . 6 % . Company s bonds have an 1 1 % coupon rate interest is payable annually,...
-
As compared to operating company IPOs, SPAC IPOs are typically a slower process. Group of answer choices True False 2. What is the primary purpose of a SPAC? Group of answer choices To take a company...
-
A skier with a mass of 73 kg is sliding down a snowy slope that makes an angle of 40.0 degrees with the horizontal. Find the coefficient of kinetic friction if the friction is known to be 57.0 N. Mk=...
-
Murthy wondered whether some biases had unconsciously crept into the decision of Teladoc's management to write down $6.6 billion of goodwill. Appendix 1 of the case lists common managerial biases....
-
What is the area and uncertainty in area of one side of a rectangular wooden board that has a length of ( 2 1 . 4 \ pm 0 . 4 ) cm and a width of ( 9 . 8 \ pm 0 . 1 ) cm ? ( Give your answers in cm 2...
-
Briefly explain in what sense a country can run a balance of payments surplus or deficit. How would a country finance a balance of payments deficit in this sense? What important differences are there...
-
Refer to Exercise 8.S.I. Construct a scatterplot of the data. Does the appearance of the scatterplot indicate that the pairing was effective? Explain. Exercise 8.S.I. A volunteer working at an animal...
-
Most South American nations produce about half of their electricity by burning natural gas to generate electricpower. Natural gas is easier to find, extract, and put to use in Brazil than in...
-
The first step in the biological degradation of histidine is formation of a 4-methylidcneirnidazol-5-one (MIO) by Cyclization of a segment of the pep- tide chain in the histidine ammonia lyase...
-
The first step in the biological degradation of lysine is reductive amination with ?-ketoglutarate to give saccharopine. Nicotinamide adenine dinucleotide phosphate (NADPH), a relative of NADH, is...
-
Carnauba wax, used in floor and furniture polishes, contains an ester of a C32 straight-chain alcohol with a C20 straight-chain carboxylic acid. Draw its structure.
-
As a HR manager identify and assess the drivers of high and low employment engagement. Support your arguments with specific examples. Please leave reference.
-
How does digital technology influence both the issues of credentialism and social/cultural reproduction? Does the technology help students? Does it hurt students? Please defend your answers using...
-
Using the four-drive motivation theory, as described by McShane, discuss how this experience is impacting Sophia's motivation ?

Study smarter with the SolutionInn App


