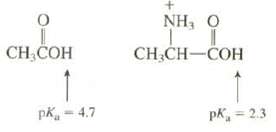
The pKa of the carboxylic acid group of acetic acid is 4.7. The pKa of the carboxylic
Question:
The pKa of the carboxylic acid group of acetic acid is 4.7. The pKa of the carboxylic acid group of the conjugate acid of the amino acid alanine is 2.3. Explain the difference in these pKa values.
Transcribed Image Text:
CH,COH 1 pk = 4.7 NH, Ο I CH₂CH-COH pk = 2.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
The NH 3 is an inductiv...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In glycine, the carboxylic acid group has Ka = 4.3 X 10-3 and the amino group has Kb = 6.0 X 10-5. Use these equilibrium constant values to calculate the equilibrium constants for the following. a....
-
Acid HA has pKa = 20; acid HB has pKa = 10. (a) Which is the stronger acid? (b) Will an acid-base reaction with an equilibrium lying to the right take place if Na+A- s added to HB? Explain your...
-
Acetic acid is continuously extracted from a 3 wt% dilute solution in water with a solvent of isopropyl ether in a mixer-settler unit. The flow rates of the feed and solvent are 12,400 and 24,000...
-
Simplify the expression 2x + (x + 1) into a single x + 1 fraction. The numerator of your answer is: The denominator of your answer is:
-
Describe the factors that managers may consider when assessing the significance of a variance. Why do we need to assess the significance of variance?
-
What is the immigration puzzle described in Chapter 11? How does the expanded model of economic growth described in equation (12.8) help to solve this puzzle?
-
Estimate the energy that a runner expends to overcome aerodynamic drag while running a complete marathon race. This expenditure of energy is equivalent to climbing a hill of what height? List all...
-
Given the following information, draw the project network. Compute the early, late, and slack times for the project network. Which activities on the critical path have only the start or finish of the...
-
The Olney Corporation provided the following information to their CPA. The accountant was asked to help them answer the questions listed below. Sales $ 274,000 Variable expenses 225,000 Contribution...
-
A Global private bank is aggressively looking to leverage technology to improve customer experience and reduce operational costs. Over the last few years, it has tied up with at least five startups...
-
Explain which nitrogen in the ring of the amino acid histidine is the stronger base: N- N-H CH + HN CHCO, Histidine
-
There are two isomeric conjugate acids that produce the following base. Show the structure of each and explain how they produce the same base upon loss of a proton. :0: CH3-C=CH
-
A coal-burning steam power plant produces a net power of 300 MW with an overall thermal efficiency of 32 percent. The actual gravimetric air-fuel ratio in the furnace is calculated to be 12 kg air/kg...
-
If a nurse knows that a piece of equipment is defective, should the nurse attempt to fix the problem? Discuss your answer.
-
Discuss the objectives and classification of criminal offenses.
-
Discuss the criminal trial process.
-
Explain the elements of a contract.
-
Discuss the distinctions among negligent torts, intentional torts, and strict liability.
-
Which type of circuit is favored for operating several electrical devices, each independently of the otherseries or parallel? Defend your answer.
-
What is a manufacturing system?
-
If |x 4| < 0.1 and |y 7| < 0.2, use the Triangle Inequality to estimate |(x + y) 11|.
-
How would you prepare 1-phenyl-2-butanone, C6H5CH2COCH2CH3, from benzyl bromide, C6H5CH2Br? More than one step is required.
-
Cyclopentanecarboxylic acid and 4-hydroxycyclohexanone have the same formula (C6H10O2), and both contain an OH and C = O group. How could you distinguish between them by IR spectroscopy?
-
How could you distinguish between the isomers Cyclopentanecarboxylic acid and 4-hydroxycyclohexanone by 1H and 13C NMR spectroscopy?
-
Donna wants to buy a savings bond for her new born niece. The face value of the savings bond is $500, the amount the niece would receive in twenty years(future value).The government is currently...
-
Lets take a $25,000 loan over six years at 8% APR once again, but this time well assume it is for a car loan that offers 0% financing for the first two years of the loan or 8% financing over six...
-
Calculate the following NPV. Calculate the discount rate (Rf=3%, B=1.0, Excess Return=10.2%) and apply to the following cash flows: CF0=$100,000 CF1=$20,000 CF2=$50,000 CF3=$45,000 CF4=$65,000 Show...

Study smarter with the SolutionInn App


