The product of the following reaction has a broad absorption at 3330 cm -1 in its IR
Question:
The product of the following reaction has a broad absorption at 3330 cm-1 in its IR spectrum. Its 13C-NMR spectrum shows absorptions at 70(C), 34 (CH2), 30 (CH3), and 15 (CH3). Suggest a structure for this compound.
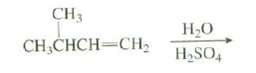
Transcribed Image Text:
CH3 Н-О H,SO, CH,CHCH=CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Because the product has a C ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many signals would the product of the following reaction show in these spectra a. Its 1H NMR spectrum b. Its 13C NMR spectrum 1. excess CH3MgBr 32. H30 CH CCH,CH,COCH
-
A compound A has a strong, broad IR absorption at 3200-3500 cm-1 and the proton NMR spectrum shown in Fig. P13.48a. Treatment of compound A with H2SO4 gives compound B, which has the NMR spectrum...
-
For each compound shown below, (1) Sketch the 13C NMR spectrum (totally decoupled, with a singlet for each type of carbon), showing approximate chemical shifts. (2) Show the multiplicity expected for...
-
7. Arrange the following nitrogen containing compounds in decreasing order of basicity NH NH -H (P) (a) S>R>Q> P NO (R) (2) (b) P>Q>S>R -H (S) (c) P>Q>R>S (d) R>Q> P > S
-
On a warm summer day in early July 2011, Ms. Smith of the executive team of X Company sat in her office preparing a request form for routine travel expense reimbursement. She attached documentation...
-
Differentiate between financial and management accounting. Give an example of how management accounting reports would be incorporated into financial accounting reports.
-
0.8859 Use the Standard Normal Table or technology to find the z-score that corresponds to the cumulative area or percentile. Table 4-Standard Normal Distribution Arca Z 0 Z .09 .08 .07 .06 .05 .04...
-
Imagine that you are trying to evaluate the economics of purchasing an automobile. You expect the car to provide annual after-tax cash benefits of $1,200 at the end of each year and assume that you...
-
The switch statement has the following structure: switch (test variable or expression) { } case value_1: case value_2: The valid types of test variable (or expression) include all of the following,...
-
The article "Effects of Bottle Closure Type on Consumer Perception of Wine Quality" (Amer. J. of Enology and Viticulture, 2007: 182-191) reported that in a sample of 106 wine consumers, 22 (20.8%)...
-
An unknown compound, A (C7H10), show four absorptions in its 13C-NMR spectrum, at 22 (CH2), 24, 124 (CH), and 126 (CH) on reaction with excess H2 and a Pt catalyst, A produces B (C7H14) B shows a...
-
Explain how absorptions may appear in the 1H-NMR spectrum of thiscompound: . Cl. CI H.
-
Show that y = x 1/2 w(2/3 x 3/2 ) is a solution of Airys differential equation y'' + 2 xy = 0, x > 0, whenever w is a solution of Bessels equation of order 1/3, that is, t 2 w'' + tw' + (t 2 1/9)w...
-
Choose any special event (not a Mega Event) and list its proper event classification, date, and location. It can be a concert, fair, festival, etc. Please keep it somewhere in the USA. Type a short...
-
Howard Company is considering three financing plans: all equity; 60 per cent equity and 40 per centdebt; and 40 per cent equity and 60 per cent debt. Total funds needed are Rs 300,000. EBIT is...
-
membership with Junktrader, which provides them advance notice of potentially attractive offers. a. Junktrader provided online advertising services for another company for $380 on account. b. On the...
-
calculate the initial concentrations at the moment of mixing before the reaction occurs of H2O2 and I-and Vitimin C for this expirement where the values given are: 2.0 mL of VitC/12 solution, 2.0 mL...
-
Research on Boeing 737 max provide a general overview of the ethical issues that occurred. Specifically, provide the following information: 1. Provide an overview of the Case (a short summary ) 2....
-
We are taking a random sample from the normal(, 2 ) distribution where we know the standard deviation = 4. Suppose that our prior experience in similar situations leads us to believe that the prior...
-
At the beginning of the year, Lam Ltd. had total assets of $800,000 and total liabilities of $500,000. Use this information to answer each of the following independent questions. (a) If Lam's total...
-
When shopping at a mall, an individual can try on an item of clothing before purchasing it to make sure it fits. When shopping online, however, the individual cannot try on the item. If it does not...
-
Which of the following alkenes can exist as c is-trans isomers? Write their structures. Build hand-held models to prove that one isomer is not superposable on the other. (a) CH2==CHCH2CH3 (b)...
-
Use VSEPR theory to predict the geometry of each of the following molecules and ions: (a) H4 (b) BeF2 (c) (d) H2S (e) BH3 (f) CF4 (g) SiF4 (h)
-
Predict the bond angles of (a) F2C==CF2 (b) CH3Ci==CCH3 (c) HCi==N
-
When owning and operating any business, whether it's a caf or a pool cleaning business, it's very important that you know exactly what your income and expenditure is. Is your business making money or...
-
Resources Unlimited Corporation was formed in 1985 through the merger of two natural pipeline companies. The result was the largest gas distribution network in the United States, with 38,000 miles of...
-
Toyota was the world's first automobile manufacturer to produce more than 10 million vehicles per year, which it has done since 2012, when it also reported the production of its 200-millionth...

Study smarter with the SolutionInn App


