The step-growth polymer nylon 6 is prepared from caprolactam. The reaction involves initial reaction of caprolactam with
Question:
The step-growth polymer nylon 6 is prepared from caprolactam. The reaction involves initial reaction of caprolactam with water to give an intermediate open?chain amino acid, followed by healing to form the polymer. Propose mechanisms for both steps, and show the structure of nylon 6.
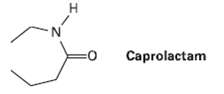
Transcribed Image Text:
H. Caprolactam
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Step 1 Water opens the caprolactam ring to form the amino acid ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Sodium reacts with water to give sodium hydroxide and gaseous hydrogen.
-
The amino acid cysteine has the structure shown: (a) A second sulfur-containing amino acid called cystine (C6H12N2O4S2) is formed when cysteine undergoes biological oxidation. Suggest a reasonable...
-
The amino acid cysteine has the structure shown:
-
Garcia Corporation has paid 60 consecutive quarterly cash dividends (15 years). The last 6 months, however, have been a cash drain on the company, as profit margins have been greatly narrowed by...
-
Select a business topic that interests you. Prepare a bibliography of at least five current magazine or newspaper articles, three books, and five online references that contain relevant information...
-
Consider Figure 1.2. a. If the name of the 'CS' (Computer Science) Department changes to 'CSSE' (Computer Science and Software Engineering) Department and the corresponding prefix for the course...
-
Describe the shape of the distribution. Use the data set, which represents the points scored by each player on the Montreal Canadiens in the 20152016 NHL season. 7507 17 10 0 19 2 18 9 5 5 26 26 0 12...
-
Air at a pressure of 1 atm and a temperature of 50C is in parallel flow over the top surface of a flat plate that is heated to a uniform temperature of 100C. The plate has a length of 0.20 m (in the...
-
A desk is made of wood with density 9 0 0 kg m - 3 . It consists of a desktop 1 2 0 cm wide, 2 cm thick and 9 0 cm deep, resting on four square wooden legs 8 0 cm long and 5 cm on the other two...
-
The T-accounts of McMahon Farm Equipment Repair follow as of May 31, 2018. Prepare McMahon Farm Equipment Repair's trial balance as of May 31, 2018. ASSETS LIABILITIES EQUITY EQUITY Contributed...
-
The following reaction, called the benzilic acid rearrangement, takes place by typical carbonyl-group reactions. Propose a mechanism (Ph =phenyl). 1. NaOH, H,0 2. H30+ Ph C-C Ph Ph Ph Benzil...
-
Qiana, a polyamide fiber with a silky texture, has the following structure. What is the monomer units used in the synthesis ofQiana? flomdoCor CH2- Qiana CICH2)6C-NH- NH-
-
Assume the same data as in Problem 5-15B with the following additional information. The hours of machine time for processing plates are 1,000 for Kate plates and 2,500 for Monroe plates. Required a....
-
A patient account with a balance of $400 was turned over to the collection agency. The collection agency was able to collect the full amount. This collection agency charges 40% of the amount...
-
What distinguishes the act of cost accounting and estimating? Will this activity ever become a science?
-
Consider the three-mass system below. Assume the following: The heavy wheel m3 rolls against the vertical wall without slipping. The forces from springs k3 and k4 and the damper c2 pass through the...
-
In 1977, Jack Grubman started work at AT&T. In 1985, he moved to PaineWebber. By 1994, he was making a million dollars a year and moved to Salomon which then became Salomon Smith Barney. During this...
-
Essential Super Market Corporation holds a 70% stake in Sandalwood Products Corporation's common stock. On January 1, 20X1, Essential purchased land for $500,000 from an independent third party. On...
-
The president of DePaul University wants to conduct a survey to determine the communitys opinion regarding campus safety. The president divides the DePaul community into three groups: resident...
-
Write a declaration for each of the following: a. A line that extends from point (60, 100) to point (30, 90) b. A rectangle that is 20 pixels wide, 100 pixels high, and has its upper-left corner at...
-
Draw an energy diagram for the molecular orbitals of period 2 diatomic molecules. Show the difference in ordering for B 2 , C 2 , and N 2 compared to O 2 , F 2 , and Ne 2 .
-
Explain why borazole (sometimes called inorganic benzene) is a very stable compound. N:N borazole
-
Although aldehydes and ketones are weak acids, their a-hydrogens are rnore than 30 pKa units more acidic than the hydrogen's of alkanes. Using polar effects and resonance effects in your argument,...
-
Although aldehydes and ketones are weak acids, their a-hydrogens are rnore than 30 pKa units more acidic than the hydrogen's of alkanes. Using polar effects and resonance effects in your argument,...
-
Use the graph above to answer the following question. If the price is set to $3, what is the producer surplus in the market? Note: enter answer with only numerical values and round up to the nearest...
-
How could your cultural models influence your ability to learn your students' funds of knowledge and utilize those funds? How could you or will you address this?
-
Bella deposits $300 per month into an account paying 6% nominal interest compounded monthly for 3 years, to be used to purchase a car. Which equation should she use to compute the price of the most...

Study smarter with the SolutionInn App


