The triene shown here reacts with 2 equivalents of maleic an hydride to yield a product with
Question:
The triene shown here reacts with 2 equivalents of maleic an hydride to yield a product with the formula C17H16O6. Predict a structure for theproduct.
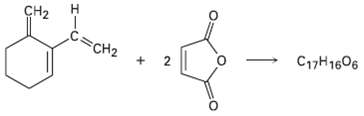
Transcribed Image Text:
CH2 C17H1606 C=CH2 2. エーU
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
The first equivalent of maleic anhydride adds to the ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In step (a), the hydrazone 8 reacts with 2 equivalents of butyl lithium (BuLi) to generate the anion 14. Propose a mechanism for this conversion. (4 marks) TBSO OBn OTBS OTPS TPSO- a. n-BuLi, THE OBn...
-
The following triene reacts with excess maleic anhydride to produce a compound with molecular formula C 14 H 12 O 6 . Draw the structure of this product (ignoring stereochemistry). Maleic anhydride...
-
Predict the major product when each reagent reacts with ethylene oxide. (a) NaOCH2CH3 (sodium ethoxide) (b) NaNH2 (sodium amide) (c) NaSPh (sodium thiophenoxide) (d) PhNH2 (aniline) (e) KCN...
-
Jen and Berry's sells ice-creams from its factory-shop in Petone. There is a managing director (Maynard Dibble), a marketing manager (Mary Salman) and a production manager (Peter Pritchard). Maynard...
-
What are some of the most significant benefits of Pay for Performance System?
-
For the RLC circuit shown in Fig. 16.59, find the complete response if v(0) = 2 V when the switch is closed. 6 1E1 2 cos 4t V (t
-
The following cost data, which include operating cost and depreciation of capital investment, pertain to Exercise 9.1. Determine by finding the total cost for each of the 14 possible sequences: (a)...
-
Belanger Ld. reports a current ratio of 2- to- 1 in its 20X2 financial statements. The statement of Financial position shows current assets of $ 2,540,500 and current liabilities of $ 1,284,000....
-
Suppose the Jenson's alpha of a portfolio is 2.5%, while the actual return is 12%. The risk-free rate is 2.5% and the expected return on the market is 12%. What is the Treynor Ratio of the portfolio?
-
The master budget at Cherrylawn Corporation at the beginning of the year was based on sales of 275,000 units with revenues of $3,300,000. Total variable costs were budgeted at $1,925,000 and fixed...
-
In light of your answer to Problem 14.43, propose a mechanism for the followingreaction: CH CH + CO2 Heat "CCH do,c, H a-Pyrone CHC-
-
The following ultraviolet absorption maxima have been measured: 1, 3-Butadiene 217 nm 2-Mcthyl-1, 3-butadicne 220 nm 1, 3-Pentadiene 223 nm 2, 3-Dimethyl- 1, 3-hutadiene 226 nm 2, 4-Hexadiene 227 nm...
-
Let X1,..., Xn be iid n(,2), 2 known, and let have a double exponential distribution, that is, () = e-|0|//(2a), a known. A Bayesian test of the hypotheses H0: 0 versus H1: > 0 will decide in...
-
Salem is willing to invest in van worth $9,000 to do transportation. If the cash inflows of a van are expected to be $3,000 annually for four years (Horizon of investment). Its annual depreciation...
-
Nick received an email from a supplier requesting a $250,000 payment for services rendered. The email requested payment through a direct deposit, which was a change from the existing payment...
-
Food sales are $82,800 and Beverage Sales are $36,900. If food cost is 32% and Beverage cost is 25%, Calculate Gross Profit.
-
Range Resources stock sells for 120. The interest rate is 3 percent. The time to expiration is nine months on all call options. The standard deviation on Noble stock is 25 percent. The interest rate...
-
Calculate E(rX), E(rY), X and Y respectively. Consider the following two stocks. Assume that the risk-free rate is 2% and agents' utility functions are U= E(rp) - 0.5Ao. Stock X currently trades at...
-
Using real-world examples, discuss the effectiveness of fiscal policy.
-
Find the work done in pumping all the oil (density S = 50 pounds per cubic foot) over the edge of a cylindrical tank that stands on one of its bases. Assume that the radius of the base is 4 feet, the...
-
What kinds of intermolecular forces are involved in solution formation?
-
Show how you would accomplish the following syntheses efficiently and in good yield. You may use any necessary reagents. (a) acetaldehyde lactic acid, CH3CH(OH)COOH (b) (c) (d) (e) (f) (g) CHPh CHO...
-
Show how you would synthesize the following derivatives from appropriate carbonyl compounds. (a) (b) (c) (d) (e) (f) CH,O OCH,
-
Predict the products formed when cyclohexanone reacts with the following reagents. (a) CH3NH2. H+ (b) Excess CH3OH. H+ (c) Hydroxylamine and weak acid (d) Ethylene glycol and p-toluenesulfonic acid...
-
You have a business worth $1,000,000 and other assets (including your home) worth an additional $750,000. What is the recommended automobile insurance policy that you should carry to be FULLY...
-
Republic Services and Waste Management company Compare the companies' long term Revenue, Gross Profit, Operating Profit) and Net Income and related growth trends. What is the average three year...
-
A young couple own a home in NJ. They would like to have Homeowner Insurance coverage for their home in NJ, in case of frozen plumping and damage form heavy snow or power surges. Which Homeowner's...

Study smarter with the SolutionInn App


