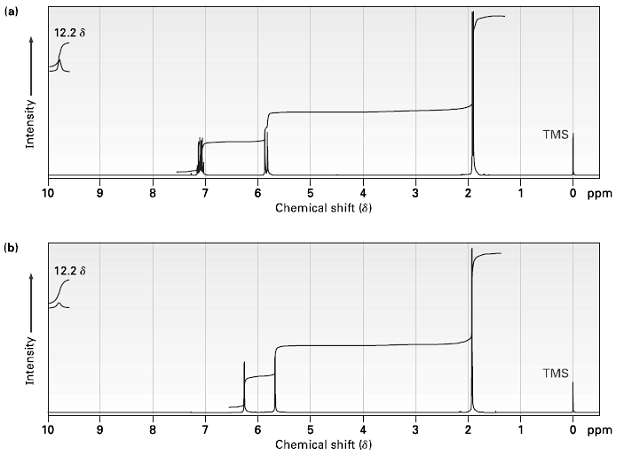
The two 1H NMR spectra shown here belong to crotonic acid (trans-CH3CH = CHCO2H) and meth acrylic
Question:
The two 1H NMR spectra shown here belong to crotonic acid (trans-CH3CH = CHCO2H) and meth acrylic acid [H2C = C (CH3) CO2H]. Which spectrum corresponds to which acid? Explain.
Transcribed Image Text:
(a) 12.2 8 TMS O ppm 10 8. 4 2 Chemical shift (8) (b) 12.2 8 TMS 10 4 O ppm Chemical shift (8) 3- -LO -to -6, Intensity Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
Both compounds contain four different kinds of protons ...View the full answer

Answered By

Ayush Mishra
I am a certified online tutor, with more than 3 years of experience in online tutoring. My tutoring subjects include: Physics, Mathematics and Mechanical engineering. I have also been awarded as best tutor for year 2019 in my previous organisation. Being a Mechanical Engineer, I love to tell the application of the concepts of science and mathematics in the real world. This help students to develop interest and makes learning fun and easy. This in turn, automatically improves their grades in the subject. I teach students to get prepared for college entry level exam. I also use to teach undergraduate students and guide them through their career aim.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H NMR spectrum shown in Figure 14.8 corresponds to one of the following compounds. Which compound is responsible for this spectrum? C CH CH3 CH3 CICH2 CH2C Br2CH CHBr2 6 5 3 2 0 8 (ppm)...
-
1H NMR spectra for two haloalkanes are shown below. Propose structures for these compounds that are consistent with the spectra. (a) C5H11C1, spectrum A; (b) C4H8Br2, spectrum B. H NMR 9 H 2 H...
-
The ketone whose 1H NMR spectrum is shown here was obtained as the product of an acetoacetic ester synthesis. What alkyl halide was used in the synthesis? 10 (ppm)
-
Acculturation is an extremely important topic in this age of globalization and multiculturalism; however, it remains an especially difficult topic to study. Peoples acculturation experiences vary...
-
In what way might doing a good job of knowledge management give a company a competitive advantage?
-
Derive a differential equation (do not solve) for the temperature distribution in a straight triangular fin. For convenience, take the coordinate axis as shown in Figure P2-83 and assume...
-
The plot in question 5 has a peak at 7,200 cpm. It appears to almost disappear when the motor power (60 Hz) is cut for the capture of the coast-down. Why?
-
Laminar flow in a narrow slit (see Fig. 2B.3).? (a) A Newtonian fluid is in laminar flow in a narrow slit formed by two parallel walls a distance 2B apart. It is understood that B p + pgh = p ? pgz...
-
A 5.2 x10 -4 V/m electric field creates a 2.0 x10 17 electrons/s current in a 1.5-mm-diameter aluminum wire. A. What is the drift speed? Express your answer using two significant figures. B. What is...
-
Your favorite college football team has had a good season so far but they need to win at least two of their last four games to qualify for a New Years Day bowl bid. Oddsmakers estimate the teams...
-
Propose a structure for a compound C4H7N that has the following IR and 1H NMR spectra: 100 80 - 60 40 - 20 - 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) TMS O ppm 10 9. 8. 3. Chemical...
-
Propose structures for carboxylic acids that show the following peaks in their 13 C NMR spectra. Assume that the kinds of carbons (1, 2, 3, or 4) have been assigned by DEPT-NMR. (a) C 7 H 12 O 2 :...
-
Determine the following indefinite integrals. Check your work by differentiation. (sin 4t - sin t/4) dt
-
John agrees to buy a second-hand fishing boat from Malcolm. They agree the price of $20,000 and John pays a deposit of $2,000. He arranges to collect the boat the next day when he has his ute with...
-
would like to develop a manual to Innovative Insurancethat includes an introduction which educates prospective customers about the function/purpose of insurance. How write a memo answering this...
-
What particular legal issue arises as a result of electronic transmission of a bill of lading? Why is this knowledge important to international traders negotiating documentary credit transactions
-
In Donna Dubinsky and Apple Computer, Inc. case, how do we make sense of how she responded to Jobs' efforts to implement a JIT system without including her relating to immunity system, power and...
-
In the following cases, what is the standard of fault that the plaintiff must prove in order to win the defamation case against the defendant? a. A national television news report says that the...
-
True or False: If the before-tax \(\mathrm{WACC}=5.4 \%\) and the income tax rate is \(40 \%\), then the after-tax \(\mathrm{WACC}=9 \%\).
-
Chloroplasts are illuminated until the levels of the Calvin cycle intermediates reach a steady state. The light is then turned off. How does the level of RuBP vary after this point?
-
Calculate the packing efficiency of the body-centered cubic unit cell. Show your work.
-
Which compound has (a) the greater boiling point? (b) the greater melting point? Explain. CH, benzene toluene
-
Into the same funnel is poured carefully 50 mL of hexane (density = 0.660 g/ml) so that the other two layers are not disturbed. The hexane forms a third layer. The funnel is stoppered and the mixture...
-
Give a general balanced reaction for The complete combustion of a cycloalkane containing one ring formula CnH2n.
-
1. How does marketing create value, and how do firms become more value driven? 2. Describe a firm's stakeholders and how they are impacted by conscious marketing.
-
Beginning inventory 100 units @ $8.00 = $ 800 Purchase # 1 200 units @ $6.00 = 1,200 Purchase # 2 100 units @ $12.00 = 1,200 Total 400 units $3,200 Ending inventory is 150 units. 1. What is cost of...
-
Sam's Ice Creams, a cottage country favourite in Northern Ontario, has launched a new range of ice creams branded as "Summer Flavours." To capitalize on the cottage and tourism season from May to...

Study smarter with the SolutionInn App


